Keep up to date with the latest research and developments from Diamond. Sign up for news on our scientific output, facility updates and plans for the future.
Related publication: Austin H. P., Allen M. D., Donohoe B. S., Rorrer N. A., Kearns F. L., Silveira R. L., Pollard B. C., Dominick G., Duman R., El Omari K., Mykhaylyk V., Wagner A., Michener W. E., Amore A., Skaf M. S., Crowley M. F., Thorne A. W., Johnson C. W., Woodcock H. L., McGeehan J. E. & Beckham G. T. Characterization and engineering of a plastic-degrading aromatic polyesterase. Proc. Natl. Acad. Sci. 115, E4350--E4357 (2018). DOI: 10.1073/pnas.1718804115
While climate change has rightly received significant global attention in recent years, we have only just awoken to the sheer magnitude of plastic pollution. Eight million tons of plastic waste enters our oceans annually, impacting the environment, wildlife, and human health. Programmes like Blue Planet II have changed our perceptions about the effects of plastic, but we currently lack effective solutions to deal with the scale of the problem.
We need to find a way to recycle plastic in a circular fashion. 93% of plastic bottles are never made into new plastic bottles, but are instead ‘downcycled’ into lower grade plastic, incinerated, or buried in landfill. Polyethylene terephthalate (PET) is one of the most common polluting plastics, and is used for single-use plastic bottles, textiles and carpets.
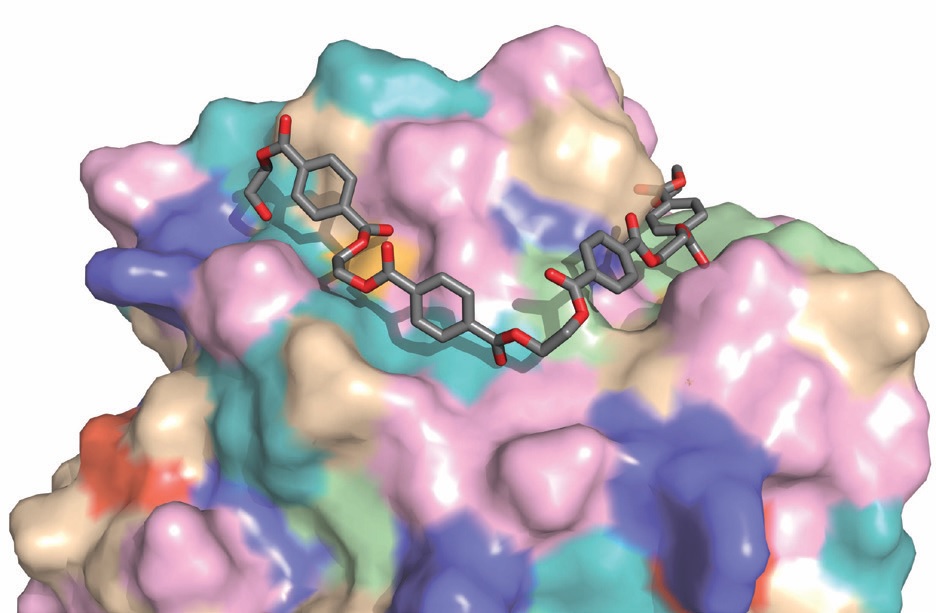
Biological enzymes have the potential to break down solid PET into its original building block monomers, and an international team of researchers used the state-of-the-art Long-Wavelength Macromolecular Crystallography (MX) beamline (I23) to investigate the 3D structure of a newly-discovered PETase enzyme to see how it works, and how to make it more efficient.
They discovered that the PETase enzyme is closely related to a cutinase enzyme produced by bacteria to digest the protective natural polyester coating on plant leaves. The 3D structure allowed them to design an improved enzyme that can digest PET faster, which offers great potential for future engineering of this enzyme for industrial recycling applications.

Plastic pollution has reached alarming levels in the environment, particularly in our oceans. From documentary programmes such as Blue Planet II, through to media from around the globe, the sheer scale of the problem is now receiving the attention that it deserves. Many plastics were only patented in the 1940s, but have gained such traction in our everyday lives that their production has reached truly staggering rates. The plastics industry is worth $1 trillion, and in the UK it represents our second largest manufacturing sector. Few could have predicted that in only a few decades huge plastic waste patches would be found floating in our oceans, and once pristine beaches would be contaminated all over the world (Fig. 1). Recent studies indicate the pervasive nature of these materials, and plastics have now been discovered in our polar sea ice and in the deepest parts of our oceans. The flow of plastic into the environment continues to grow, and it is now with some urgency that we need to find sustainable alternatives and reduce our reliance on plastics for single-use applications.
Plastics have incredible properties and have revolutionised many industries. They are cheap to manufacture, strong, light, transparent, waterproof, inert, and can be moulded into highly complex solids. From films to the ubiquitous fizzy drink bottle, they protect and extend the lifetime of many products. So why then have they created such a global scourge? It is the very properties that the chemists were asked to create, low cost and durability, that have had unforeseen consequences. To understand why, we need to look at the nature of common plastics.

Plastic polymers such as polyethylene terephthalate, or PET, are made from simple monomer building blocks that are linked together via ester bonds, hence the name polyesters. Most monomers are extracted from petroleum, although more recent developments have seen building blocks produced directly from plants. Petroleum based monomers are very cheap because of low oil prices, and it is therefore often cheaper to manufacture virgin-PET than to use recycled material. PET also suffers from down-cycling, and in the case of single-use plastic bottles, only a small percentage will be turned back into new plastic bottles. A reduction in chemical properties results in most recycled PET being used for clothing fibres, eventually reaching an end-of-cycle use as fillings or carpet. From these low value products, there is little financial incentive to recycle further and much ends up in landfill or is incinerated. While burning plastic can be used to generate electricity, this practice contributes to greenhouse gases with the release of carbon dioxide. Of course, any recycling process can only take place when there is a proper infrastructure for the collection and treatment of waste; infrastructure that is severely lacking in many parts of the world. The consequence is that much of the plastic that is not dumped into landfill or incinerated will eventually find its way into rivers and oceans, feeding the already giant plastic-waste islands from the gyres created by global ocean currents. We must reduce our reliance on single-use plastic, and develop better collection and recycling methods that are truly circular and sustainable.
In 2016, a team of scientists in Japan published an exciting research paper describing the discovery and isolation of a strain of bacteria, Ideonalla sakaiensis, which has the remarkable ability to survive on a diet of PET1. It was shown that these bacteria secreted two enzymes that can break apart the ester bonds holding the PET monomers together, providing a food and energy source for the growing organisms. This study highlights the diversity of bacteria and shows how they can adapt to new substrates, even man-made materials. Given that PET has only been in the environment for around 60 years, it is incredible that these bacteria have evolved to tackle this difficult material in such a short timeframe. The big questions for us were: how did these enzymes evolve, how do they work, and can we utilise them as part of a recycling solution?
Diamond Light Source is the UK's national synchrotron science facility, located at the Harwell Science and Innovation Campus in Oxfordshire.
Copyright © 2022 Diamond Light Source
Diamond Light Source Ltd
Diamond House
Harwell Science & Innovation Campus
Didcot
Oxfordshire
OX11 0DE
Diamond Light Source® and the Diamond logo are registered trademarks of Diamond Light Source Ltd
Registered in England and Wales at Diamond House, Harwell Science and Innovation Campus, Didcot, Oxfordshire, OX11 0DE, United Kingdom. Company number: 4375679. VAT number: 287 461 957. Economic Operators Registration and Identification (EORI) number: GB287461957003.