Keep up to date with the latest research and developments from Diamond. Sign up for news on our scientific output, facility updates and plans for the future.
The structure of a photosynthetic protein complex that absorbs infrared light in aquatic bacteria has been determined for the first time, using cryo-electron microscopy (cryo-EM) at eBIC. The work outlined in the journal Nature, showed every component of the photosynthetic complex to an impressive 2.9 Å resolution.
In photosynthesis, light harvesting (LH) complexes funnel absorbed solar energy to reaction centre (RC) complexes, which transiently trap and store the harvested energy in the form of a charge separated state. Thereafter, a series of electron and proton transfers within the RC converts a quinone acceptor to its reduced form, a quinol. Plants and algae usually absorb light energy at wavelengths of up to 700 nm, but one particular phototrophic bacterium Blastochloris (Blc.) viridis can absorb and utilise wavelengths far greater than these. Remarkably, these bacteria have adapted RCLH complexes with the capacity to absorb energy above 1000 nm – near infrared light that is inaccessible to other photosynthetic organisms.
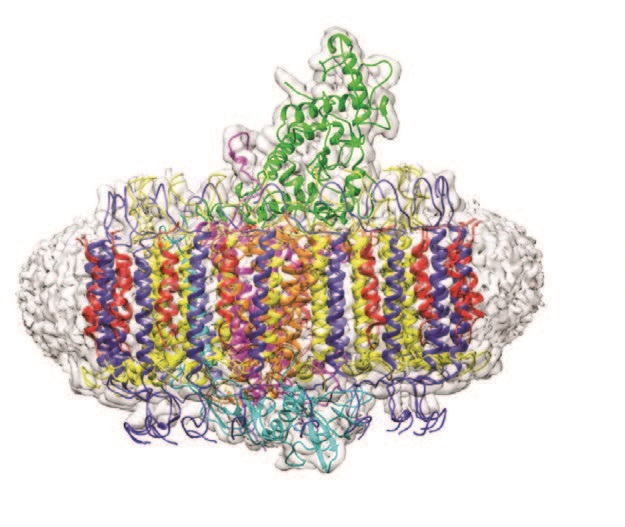
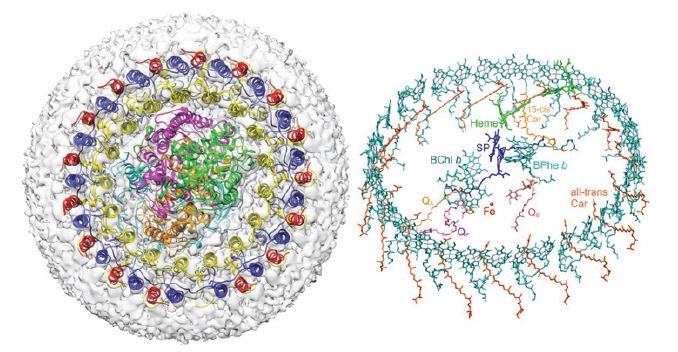
The central RC component of the Blc. viridis photosynthetic complex was the first ever membrane protein structure to be successfully determined by X-ray crystallography which resulted in the awarding of the Nobel prize in 1985 to Deisenhofer, Huber and Michel1,2. However, the structure of the LH1-RC complex in its entirety had eluded scientists until now. To understand the structural basis of the extraordinary infrared absorption, researchers from the University of Sheffield collaborated with scientists at eBIC and utilised Titan Krios I to determine the structure of the LH1-RC complex. The resulting 2.9 Å resolution structure revealed the positions of all bacteriochlorophyll, carotenoid, haem and quinone cofactors, as well as the protein-protein interactions that form the structural basis for infrared light absorption, chargetrapping electron transfers and export of the quinol product.
Plants and algae use chlorophyll a to absorb solar energy at wavelengths up to 700 nm. However, it has been known for decades that some phototrophic bacteria can make use of other, lower energy regions of the solar spectrum, by absorbing light in the 750–900 nm region. Most of these bacteria use bacteriochlorophyll a, which absorbs at around 770 nm in methanol, and their LH1-RC complexes red-shift the absorption maximum to 870–880 nm. Blc. viridis uses bacteriochlorophyll b (BChl b) which, when solubilised in methanol, absorbs at 795 nm, and remarkably, achieves a red shift of 220 nm from the pigments, enabling photosynthesis to occupy a spectral niche above 1000 nm.
The application of cryo-electron microscopy (cryo-EM) was crucial to understand the whole intact complex and its role in photosynthetic growth as the reaction centre alone cannot harvest sufficient light. The LH1 ring that surrounds the RC and co-ordinates a bacteriochlorophyll array harvests and delivers captured light energy extremely efficiently to the RC special pair. This process is illustrated in fine detail in the published structure.
The cryo-EM structure of the Blc. viridis LH1-RC complex showed that the organism employs a range of strategies to allow it to harvest light at 1000 nm. The presence of BChl b alone is insufficient to explain the absorption and the high-resolution structure indicated a number of additional contributory factors. As has been demonstrated in related organisms by site-specific mutagenesis, H-bonding between BChl b and LH1 Trp residues is likely responsible for a component of the red shift3. Additional contributions are due to the size, stiffness and extensive cooperativity within the complex between the proteins and co-ordinated pigments. The Blc. viridis LH1 complex also contains a novel gamma subunit and this enables a triple ring structure which further rigidifies the entire array thereby promoting the red shift.
The product of energy trapping is a quinol molecule, which must escape the confines of the LH1 ring that surrounds the RC and move into the quinone pool for subsequent utilisation by the cytochrome bc1 complex4. The Blc. viridis LH1-RC structure illustrates how quinols may traverse the surrounding LH1 ring. A small pore is apparent in the LH1 ring, near a binding pocket, which houses a quinol, designated QP. This binding pocket provides a folding template that imposes a compact conformation and a suitable orientation on QP prior to its entry to the pore in the LH1 ring. Thus, the RC-LH1 complex of Blc. viridis reveals a new strategy for fostering quinol/quinone exchange across an LH1 ring.
The structural insights gained from this study could enable near infrared light absorption to be engineered into biosynthetic, bioinspired or biohybrid photosynthetic structures. In particular, bacteria could be adapted to have a wide input of solar energy with applications for novel solar cell technologies.
The team will continue looking at this bacterium and they plan to apply genetic engineering to remove the critical outer ring to see if they can force the complex to absorb light towards higher energy wavelengths. Building on the success of this structural study at eBIC they will also explore other photosynthetic systems using cryo-EM.
Diamond Light Source is the UK's national synchrotron science facility, located at the Harwell Science and Innovation Campus in Oxfordshire.
Copyright © 2022 Diamond Light Source
Diamond Light Source Ltd
Diamond House
Harwell Science & Innovation Campus
Didcot
Oxfordshire
OX11 0DE
Diamond Light Source® and the Diamond logo are registered trademarks of Diamond Light Source Ltd
Registered in England and Wales at Diamond House, Harwell Science and Innovation Campus, Didcot, Oxfordshire, OX11 0DE, United Kingdom. Company number: 4375679. VAT number: 287 461 957. Economic Operators Registration and Identification (EORI) number: GB287461957003.