Diamond helps scientists visualise an enzyme from wood-eating gribble that could help turn waste into biofuel
Jun 3, 2013
Jun 3, 2013
Scientists have discovered a new enzyme that could prove an important step in the quest to turn waste (such as paper, scrap wood and straw) into liquid fuel. To do this they turned to the destructive power of tiny marine wood-borers called ‘gribble’, which have been known to destroy seaside piers, and the intense X-rays available at Diamond.

Gribble are of interest as they are voracious consumers of wood and have all the enzymes needed for its digestion. The enzymes attach to a long chain of complex sugars and chop off small soluble molecules that can be easily digested or fermented. The researchers identified a cellulase (an enzyme that converts cellulose into glucose) from gribble that has some unusual properties and used the latest imaging technology to understand more about it.
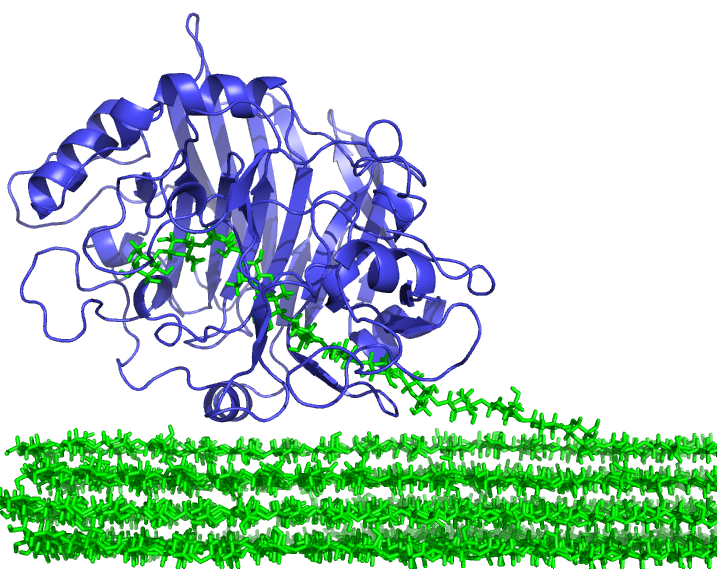
The 3D X-ray structure allows scientists to see inside the enzyme
and reveals how it binds and digests cellulose chains.
This information will help the researchers to design more robust enzymes for industrial applications. While similar cellulases have been found in wood-degrading fungi, the enzyme from gribble shows some important differences. In particular, the gribble cellulase is extremely resistant to aggressive chemical environments and can work in conditions seven times saltier than sea water. Being robust in difficult environments means that the enzymes can last much longer when working under industrial conditions and so less enzyme will be needed.
Professor McQueen-Mason explained: “This is the first functionally characterised animal enzyme of this type and provides us with a previously undiscovered picture of how they work.
“While this enzyme looks superficially similar to equivalent ones from fungi, closer inspection highlights structural differences that give it special features, for example, the enzyme has an extremely acidic surface and we believe that this is one of the features that contributes to its robustness.”
The ultimate aim is to reproduce the effect of this enzyme on an industrial scale. Rather than trying to get the cellulase from gribble, the team have transferred the genetic blueprint of this enzyme to an industrial microbe that can produce it in large quantities, in the same way that enzymes for biological washing detergents are made. By doing this they hope to cut the costs of turning woody materials into biofuels.
Professor McQueen-Mason added: “The robust nature of the enzymes makes it compatible for use in conjunction with sea water, which would lower the costs of processing. Lowering the cost of enzymes is seen as critical for making biofuels from woody materials cost effective. Its robustness would also give the enzymes a longer working life and allow it to be recovered and re-used during processing.”
The work is part of the BBSRC Sustainable Bioenergy Centre (BSBEC), a £24M investment that brings together six world-class research programmes to develop the UK's bioenergy research capacity. Funding from a BBSRC USA Partnering Award was instrumental in forming a highly synergistic collaboration with the US DOE funded research team at NREL.
Paper: Structural characterization of the first marine animal Family 7 cellobiohydrolase suggests a mechanism of cellulase salt tolerance.
Link to the PNAS paper: http://www.pnas.org/cgi/doi/10.1073/pnas.1301502110
Diamond Light Source is the UK's national synchrotron science facility, located at the Harwell Science and Innovation Campus in Oxfordshire.
Diamond Light Source Ltd
Diamond House
Harwell Science & Innovation Campus
Didcot
Oxfordshire
OX11 0DE
Copyright © Diamond Light Source. Diamond Light Source® and the Diamond logo are registered trademarks of Diamond Light Source Ltd
Registered in England and Wales at Diamond House, Harwell Science and Innovation Campus, Didcot, Oxfordshire, OX11 0DE, United Kingdom. Company number: 4375679. VAT number: 287 461 957. Economic Operators Registration and Identification (EORI) number: GB287461957003.