Keep up to date with the latest research and developments from Diamond. Sign up for news on our scientific output, facility updates and plans for the future.
Related publication: Chen Z., Vorobyeva E., Mitchell S., Fako E., Ortuño M. A., López N., Collins S. M., Midgley P. A., Richard S., Vilé G. & Pérez-Ramírez J. A heterogeneous single-atom palladium catalyst surpassing homogeneous systems for Suzuki coupling. Nat. Nanotechnol. 13, 702–707 (2018). DOI: 10.1038/s41565-018-0167-2
The chemical industry produces a tremendous amount of waste. Often this arises from the need to separate the product from a solution containing by-products and a dissolved catalyst. 85% of all industrial chemical processes use catalysts, chemicals that speed up a reaction but do not get used up in the process, and they are often expensive metals such as palladium.
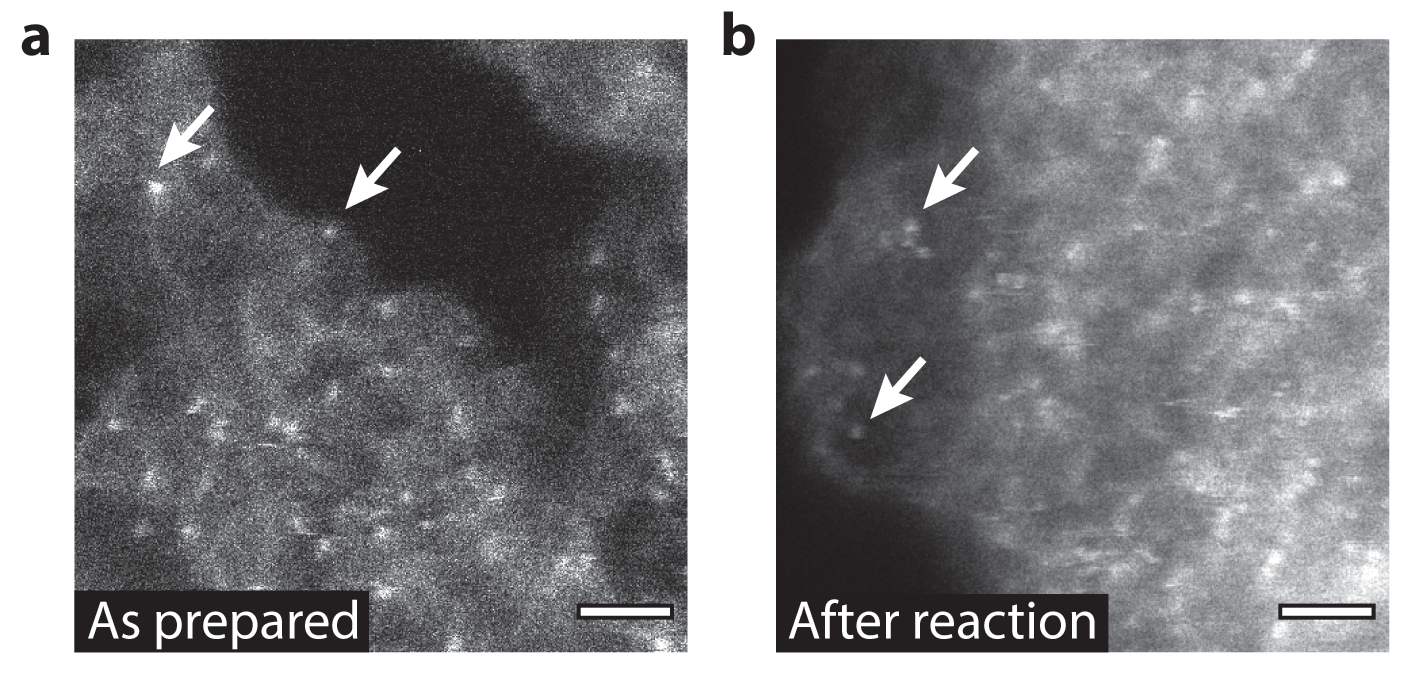
An international team of researchers sought an efficient solid catalyst that would avoid waste. They investigated a material designed to contain individual atoms of palladium supported by carbon nitride, which showed improved performance in a reaction to produce new carbon-carbon bonds.
Industrial production of fine chemicals depends critically on catalysis. In fact, 85% of all industrial processes in the chemical industries are catalytic processes1. Catalysts operate by reducing activation energies in key mechanistic steps during a chemical reaction, leading to increases in the rate of reaction at accessible temperatures. The catalyst, often relying on a transition metal species for carrying out key steps, is not used up during the reaction, and a relatively small quantity of the catalyst can participate in reactions, in principle, many times over to convert starting materials to a specific product.
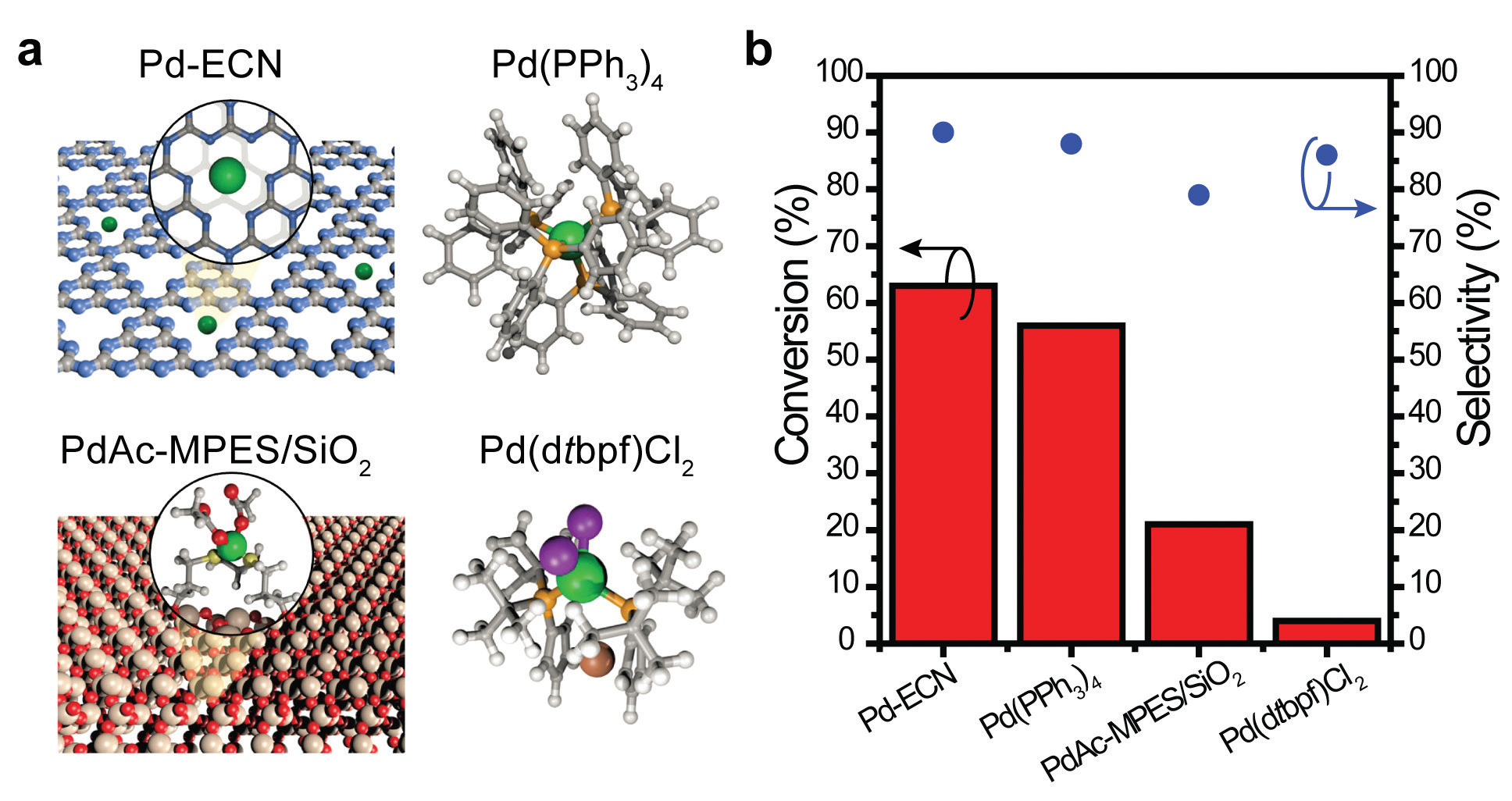
An alternative to homogeneous catalysts, and those heterogeneous designs anchoring Pd complexes to an inorganic support, has emerged with the development of single atom catalysts on carbon and nitrogen derived materials, such as graphitic carbon nitride4. Graphitic carbon nitride is a polymeric material consisting of sheets of carbon and nitrogen atoms analogously to graphene. Conceptually, the ability of graphitic carbon nitride to form bonds with bare, single Pd atoms suggests a design chemically very similar to the coordination environment of the most active homogeneous Pd cross-coupling catalysts.
Dr Sean M Collins, Department of Materials Science and Metallurgy, University of Cambridge, smc204@cam.ac.uk
Diamond Light Source is the UK's national synchrotron science facility, located at the Harwell Science and Innovation Campus in Oxfordshire.
Copyright © 2022 Diamond Light Source
Diamond Light Source Ltd
Diamond House
Harwell Science & Innovation Campus
Didcot
Oxfordshire
OX11 0DE
Diamond Light Source® and the Diamond logo are registered trademarks of Diamond Light Source Ltd
Registered in England and Wales at Diamond House, Harwell Science and Innovation Campus, Didcot, Oxfordshire, OX11 0DE, United Kingdom. Company number: 4375679. VAT number: 287 461 957. Economic Operators Registration and Identification (EORI) number: GB287461957003.