Keep up to date with the latest research and developments from Diamond. Sign up for news on our scientific output, facility updates and plans for the future.
Hexaferrites are an important class of magnetic oxides with applications in data storage and electronics, and have been the subject of numerous studies since their discovery in the 1950s. They are iron (III)-based oxides with highly modular crystal structures, formed of building blocks that can be stacked in different sequences to form a large variety of unique structures. The large size of the resulting structures makes it difficult to analyse them with atomic resolution, but the important functional properties of the hexaferrite family led a team of researchers to investigate the single-crystal growth and structural determination of very large unit cell mixed-layered hexiferrite materials.
The team grew a series of complex hexaferrite crystals, and solved their atomic-level crystal structure using high-resolution synchrotron X-ray diffraction, electron diffraction, and imaging methods, and their physical characterisation by magnetometry. X-ray diffraction data from the Small-Molecule Single-Crystal Diffraction beamline (I19) allowed the team to successfully determine the single-crystal structures of nine hexaferrite materials, all with very large unit cell parameters. The structures include a new hexaferrite stacking sequence, with the longest lattice parameter of any hexaferrite to have its structure fully determined.
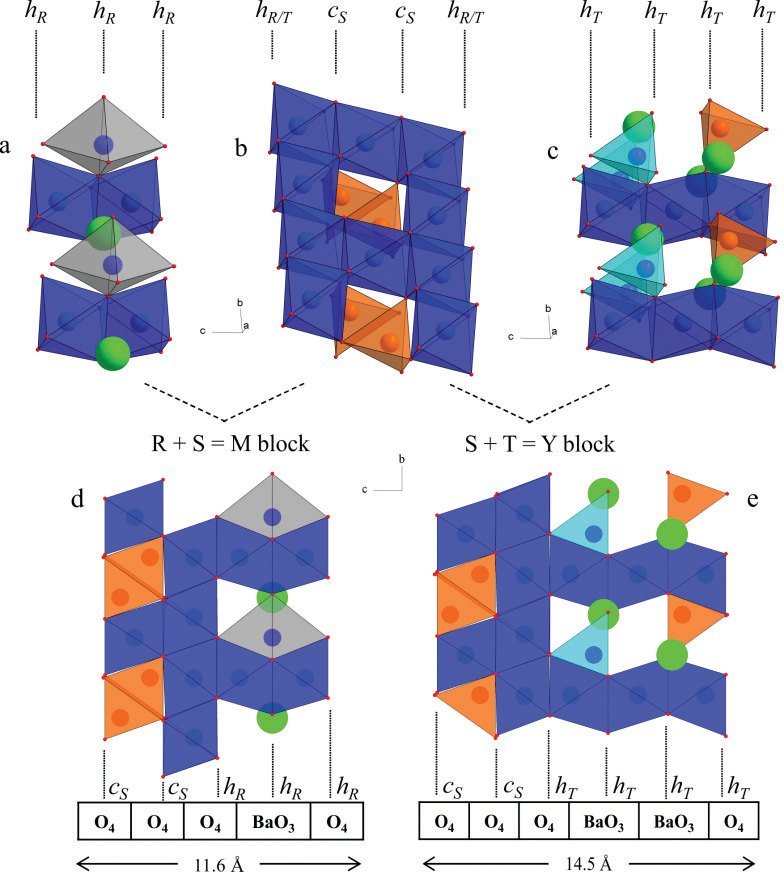
Hexaferrites are a class of iron (III)-based oxides with hexagonal, trigonal, or rhombohedral lattices, and have been extensively studied for their applications as magnets, particularly in recording and data storage devices, as well as in electrical components such as antennas1-4. All hexaferrites are ferrimagnetic, having high magnetic ordering temperatures due to localised Fe3+ cations, and strong Fe3+–O–Fe3+ antiferromagnetic superexchange interactions. The family of hexaferrites contains many varieties; the most studied being the BaFe12O19 M-type lattice, followed by BaM2Fe16O27 (W-type), Ba2M2Fe12O22 (Y-type), Ba3M2Fe24O41 (Z-type), Ba2M2Fe28O46 (X-type), and Ba4M2Fe36O60 (U-type) where typically M = Co2+, Zn2+, Fe2+, Mg2+, or Mn2+5. To add further complexity to this fascinating system, each family contains a subgroup of more complex mixed-layer structures formed from the regular stacking of M and Y unit blocks, the MpYn series, giving rise to a large number of different stacking sequences with hexagonal c parameters up to 1577 Å. These M and Y structural unit blocks are themselves built from the R, S, and T blocks (Fig. 1), which are distinguished by the stacking of their close-packed oxygen layers. In the R and T sub-blocks, the oxygen layers are in hexagonal close packing while they are in cubic close packing in the S sub-blocks. Two types of oxygen layers are present: one with a barium substitution (the {BaO3} layers), and one without (the {O4} layers).
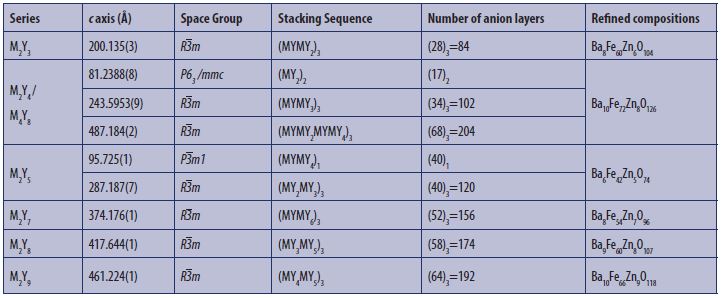 Table 1: Synthesis and single-crystal growth of target hexaferrites in this study. The level of complexity of the mixed layered compounds required the use of (N)x nomenclature where N refers to the number of anion layers of the stacking sequence, and x to the x-fold repeat of this stacking sequence within a complete unit cell.
Table 1: Synthesis and single-crystal growth of target hexaferrites in this study. The level of complexity of the mixed layered compounds required the use of (N)x nomenclature where N refers to the number of anion layers of the stacking sequence, and x to the x-fold repeat of this stacking sequence within a complete unit cell.
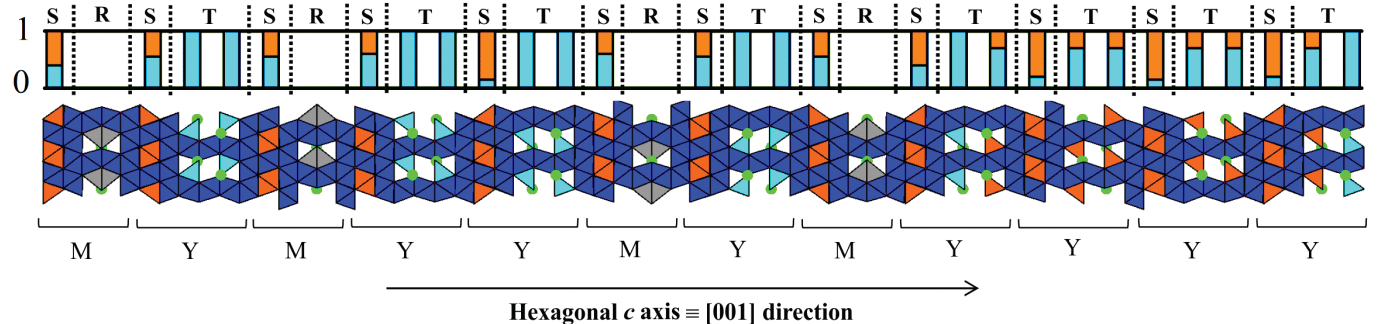
The availability of modern characterisation methods opens the possibility of accessing new complex stacking sequences, and associated properties, that lie beyond those that could be studied and understood with previously available tools. Our recent work at I19 focussed on mixed-layer hexaferrites constructed from different ratios and permutations of M- and Y- blocks, and we have now reported full structural refinement of nine mixed-layer materials belonging to the M2Y3, M2Y4/M4Y8, M2Y5, M2Y7, M2Y8, M2Y9 series (Table 1) from single-crystals in the Ba-Fe-Zn-O system. To aid in our characterisation, we determined their structures crystallographically, combining transmission electron microscopy techniques (electron diffraction and high-resolution imaging) with single-crystal X-ray diffraction data collected on I19 in order to refine complete structural models. The single-crystal X-ray refinement of the several previously reported hexaferrite systems allowed us to propose a model for the observed cation ordering within the entire MY hexaferrite series based on electrostatic potential calculations enabled by the precisely determined structures. The ability to handle large cells on I19 allowed us to realise a new stacking sequence within the M4Y8 series, with a complex structure that corresponds to the largest repeat known for an oxide material within the Inorganic Crystal Structure Database (ICSD).
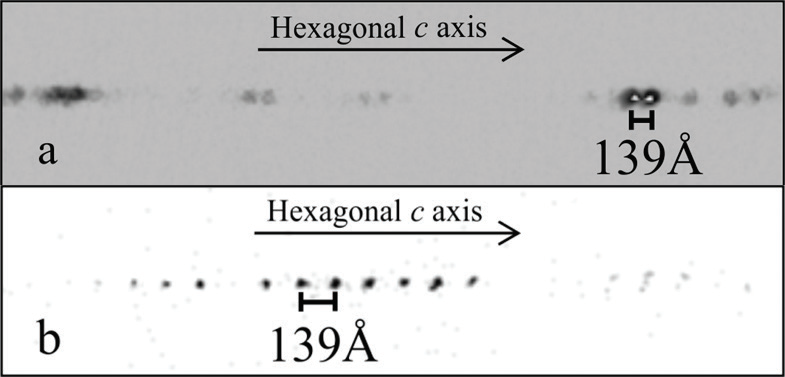
The structural refinement of the large unit cells observed in this study was indeed challenging due in part to the long hexagonal repeats, where reflection overlap was prevalent when using the in-house diffractometers (Fig. 2a). Thus, to solve these structures, synchrotron instrumentation was required, enabling the use of a 300 mm detector distance, and the acquisition of well-separated reflections (Fig. 2b). I19 data collections were essential, and allowed the determination of these mixed-layer structures. The mixed-layer structural models were then solved and refined with synchrotron X-ray diffraction data.
References:
Diamond Light Source is the UK's national synchrotron science facility, located at the Harwell Science and Innovation Campus in Oxfordshire.
Copyright © 2022 Diamond Light Source
Diamond Light Source Ltd
Diamond House
Harwell Science & Innovation Campus
Didcot
Oxfordshire
OX11 0DE
Diamond Light Source® and the Diamond logo are registered trademarks of Diamond Light Source Ltd
Registered in England and Wales at Diamond House, Harwell Science and Innovation Campus, Didcot, Oxfordshire, OX11 0DE, United Kingdom. Company number: 4375679. VAT number: 287 461 957. Economic Operators Registration and Identification (EORI) number: GB287461957003.