Keep up to date with the latest research and developments from Diamond. Sign up for news on our scientific output, facility updates and plans for the future.
Aerosols – tiny particles in the air – are key to the formation of clouds, as all cloud droplets form when water droplets condense around aerosols. If the chemical properties of aerosols are modified, this can affect the size and number of droplets within a cloud, and hence how much light the cloud reflects and how much rain falls. A small chemical change can cause a climatic difference, and although this process has a significant effect on Earth’s climate, it is not well understood.
Atmospheric aerosols critically affect the Earth’s climate by directly scattering and absorbing solar radiation and indirectly through their involvement in cloud formation and their influence on cloud radiative properties1. Numerous aerosols are coated in a thin organic film, which affects their physical properties and therefore their contribution to the Earth’s climate2. As the atmosphere is an oxidising medium, it is likely that an aerosol will be oxidised in its lifetime, which will also affect the physical properties of the aerosol such as its ability to uptake water and whether it will act as cloud condensation nuclei (CCN), the precursor to a cloud droplet. Therefore, it is important to study oxidation reactions of atmospheric aerosols coated in a thin film to better understand aerosol contribution to cloud formation and cloud radiative properties and thus the Earth’s climate.
X-ray reflectivity studies were performed on monolayer films of atmospheric or sea-water material at the air-water interface formed on a Langmuir trough. A custom trough was set up on an anti-vibration table on the I07 beamline and reflectivity of the film was monitored before and during addition of an oxidant.
For experiments conducted using ozone as the oxidant, ozone was generated using an ozoniser and the trough was contained in a sealed chemically inert plastic bag. Kapton windows allowed X-rays to pass into the bag. For aqueous phase radical experiments, hydroxyl or nitrate radicals were generated in the aqueous subphase from photolysis of a precursor solution. UV lamps were located directly above the trough along its length to evenly illuminate the subphase.
X-rays were specularly reflected from the liquid surface onto a defined region of interest on a detector and the reflectivity of the film was measured as a function of momentum transfer of the X-rays perpendicular to the interface. The X-ray data was modelled using a simple parallel layer model consisting of the thin film located between two semi-infinite media (air and the liquid subphase) as shown in Figure 1.

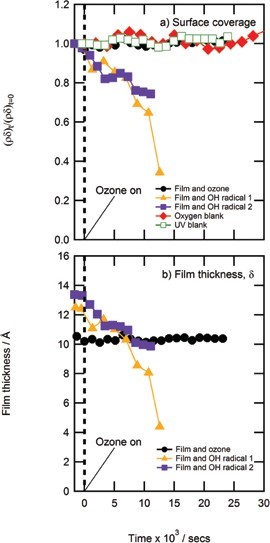
No reaction was observed between gas-phase ozone and a thin film of material extracted from atmospheric aerosol or sea-water. The amount of material at the air-water interface remained constant throughout the experiment. Owing to this surprising result, a thin film of material extracted from atmospheric aerosol was exposed to the aqueous phase hydroxyl radical and a reaction was observed. Figure 2 shows the reflectivity profile for the reaction, the reflectivity increases as shown by the direction of the arrow as time proceeds indicating a reaction. The X-ray data was modelled3 (Fig. 3), and a clear decrease in scattering length density and film thickness was observed in comparison to the constant values observed when atmospheric film material was exposed to gas-phase ozone.
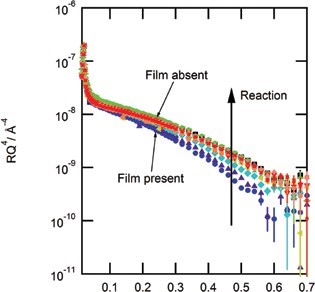
As a reaction was observed with the aqueous phase hydroxyl radical and no reaction was observed with gas-phase ozone, it suggests that the organic film material in both atmospheric and sea-water samples contains a lack of unsaturated material. Unsaturated material contains double bonds which ozone would cleave resulting in the formation of smaller molecules that would either partition to the gaseous or aqueous phase resulting in a decrease in the amount of material at the air-water interface. Aqueous radicals react by abstraction of a hydrogen atom and can therefore react with material that contains no double bonds. Similarly no reaction was observed between sea-water film material and gas-phase ozone, however, a reaction was observed (decrease in the amount of material at the air-water interface) between sea-water film material and the aqueous phase nitrate radical.
Atmospheric aerosol and sea-water samples were also analysed by mass spectrometry techniques. No unsaturated material was detected, which could mean that the concentration of unsaturated material in the samples is very low and could even be below the detection limit of the techniques, or, that the samples contain a much greater proportion of saturated material. In support of the lack of reaction observed with gas-phase ozone, a number of studies4,5 in the literature have shown that unsaturated material exists in much lower concentrations compared to saturated material in atmospheric aerosol samples.
In conclusion, organic material extracted from real atmospheric aerosol and sea-water samples forms stable films at the air-water interface. The oxidation of these films has been studied using X-ray reflectivity and shown that such films are unreactive to gaseous ozone and can be modelled as a constant amount of material at the air-water interface. It is proposed that the samples either do not contain unsaturated material or contain it in very low concentrations, which is supported by multiple studies in the literature4,5which propose that unsaturated material in atmospheric aerosol is present in very low concentrations when compared to saturated material. X-ray reflectivity has shown that a thin organic film extracted from atmospheric aerosol does react with the aqueous phase hydroxyl radical and a thin organic film extracted from sea-water does react with the aqueous phase nitrate radical. Therefore heterogeneous oxidation by gas-phase ozone may not be important in the atmosphere, whereas radical oxidation appears to be important. Thus it is recommended that future proxy studies focus on radical oxidation of saturated organics. Future studies of real aerosol samples collected at different stages throughout an aerosol lifetime are also recommended to determine whether gas-phase oxidation by ozone is important at specific times.
Jones SH, King MD, Ward AD, Rennie AR, Jones AC, Arnold T. Are organic films from atmospheric aerosol and sea water inert to oxidation by ozone at the air-water interface? Atmospheric Environment 161, 274-287, doi:10.1016/j.atmosenv.2017.04.025 (2017).
Diamond Light Source is the UK's national synchrotron science facility, located at the Harwell Science and Innovation Campus in Oxfordshire.
Copyright © 2022 Diamond Light Source
Diamond Light Source Ltd
Diamond House
Harwell Science & Innovation Campus
Didcot
Oxfordshire
OX11 0DE
Diamond Light Source® and the Diamond logo are registered trademarks of Diamond Light Source Ltd
Registered in England and Wales at Diamond House, Harwell Science and Innovation Campus, Didcot, Oxfordshire, OX11 0DE, United Kingdom. Company number: 4375679. VAT number: 287 461 957. Economic Operators Registration and Identification (EORI) number: GB287461957003.