Keep up to date with the latest research and developments from Diamond. Sign up for news on our scientific output, facility updates and plans for the future.
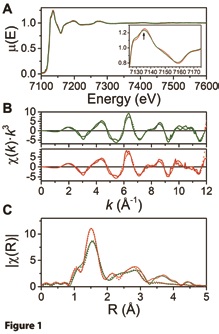
The Extended X-ray Absorption Fine Structure (EXAFS) signals and the corresponding Fourier transforms are shown in Figure 1b and Figure 1c respectively. The Fourier transforms indicate an increase in disorder for the first coordination sphere around the iron centre for the mutant species. After an initial estimation of the distances between metal and ligand using X-ray diffraction data, a cyclic refinement was performed by selecting coordinates from 10000 snapshots of Molecular Dynamics computations, which determined the lowest squared deviations in distances for the distinct scattering paths selected. Interestingly, there was no significant difference in metal-to-ligand distances in between the two species. Nevertheless, Debye-Waller factors indicated an increase of the disorder in the equatorial ligands – those belonging to the phophyrin ring – in agreement with the broadening of the 7136 eV feature in the XANES region and the decrease of intensity of the first peak of the Fourier transform.
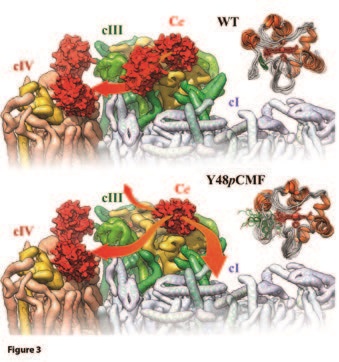
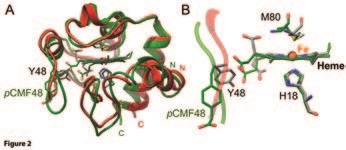
Moreno-Beltrán B, Guerra-Castellano A, Díaz-Quintana A, Del Conte R, García-Mauriño SM, Díaz-Moreno S, Gonzalez-Arzola K, Santos-Ocaña C, Velázquez-Campoy A, De la Rosa Ma, Turano P, Díaz-Moreno I. Structural Basis of Mitochondrial Dysfunction in Response to Cytochrome c Phosphorylation at Tyrosine 48. Proceedings of the National Academy of Sciences 114 (15), 3041-3050, doi:10.1073/pnas.1618008114 (2017).
Diamond Light Source is the UK's national synchrotron science facility, located at the Harwell Science and Innovation Campus in Oxfordshire.
Copyright © 2022 Diamond Light Source
Diamond Light Source Ltd
Diamond House
Harwell Science & Innovation Campus
Didcot
Oxfordshire
OX11 0DE
Diamond Light Source® and the Diamond logo are registered trademarks of Diamond Light Source Ltd
Registered in England and Wales at Diamond House, Harwell Science and Innovation Campus, Didcot, Oxfordshire, OX11 0DE, United Kingdom. Company number: 4375679. VAT number: 287 461 957. Economic Operators Registration and Identification (EORI) number: GB287461957003.