Keep up to date with the latest research and developments from Diamond. Sign up for news on our scientific output, facility updates and plans for the future.
In all cells derived from animals, plants, fungi and protozoa a single protein called UGGT acts as a quality control checkpoint that inspects secreted glycoproteins. UGGT has fascinated scientists for many years as it has the capacity to check the quality of a huge array of glycoproteins, which come in many different shapes and sizes and folds. The way in which it screens such a diverse group of proteins is poorly understood, and no structures exist of UGGT. Furthermore, UGGT is implicated in human diseases as it can ensure that glycoproteins from viruses (such as influenza, hepatitis C, HIV and Zika) are folded correctly. By fully characterising UGGT, it is hoped that an inhibitor could be developed that may act as a broad-spectrum anti-viral drug.
Using the Macromolecular Crystallography (MX) beamlines (I02, I03, I04 and I04-1) and cryo-electron microscopy (cryo-EM) at the Electron Bio-Imaging Centre (eBIC), the structure of UGGT was solved. Four distinct crystal structures of a full-length UGGT revealed a novel seven domain fold, with four thioredoxin-like domains arranged in a long arc, which terminates in two β–sandwich domains that tightly clasp the catalytic domain. Together, the cryo-EM map, the crystal structures and the biochemical data suggested that UGGT is highly flexible, a feature which enables it to check such a wide variety of glycoproteins.
A wonderfully efficient protein folding machinery in the Endoplasmic Reticulum (ER) of eukaryotic cells ensures that only correctly folded glycoproteins can exit the ER and proceed to the Golgi, and from the Golgi continue along the secretory pathway towards their individual cellular or extracellular destinations.
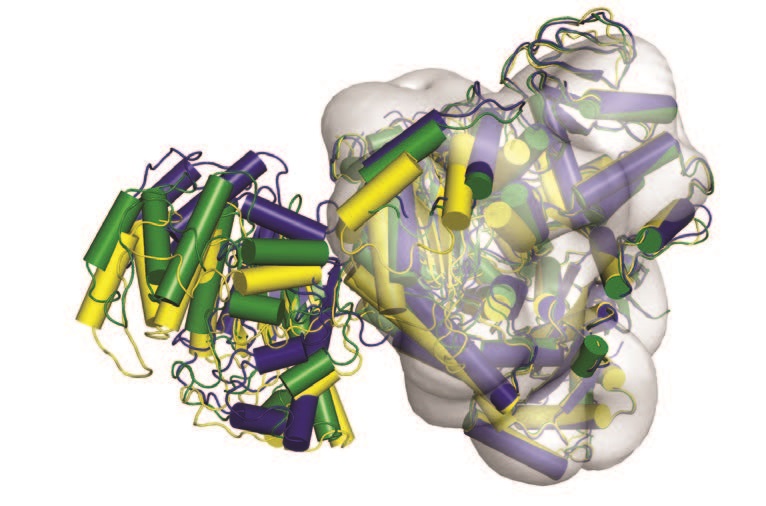
Together with the 15 Å cryo-EM map, the same UGGT crystal structures reveal pronounced inter-domain flexibility (Fig. 1). Structure-based double cysteine point mutants that engineer extra inter-domain disulfide bridges, rigidify the UGGT structure and exhibit impaired activity. Thus, UGGT enzymatic activity depends on inter-domain conformational mobility, suggesting UGGT’s intrinsic flexibility as a key requirement for the enzyme to be able to recognise and react with its many different misfolded substrates, flagging them for retention. By changing the relative orientations between its four TRXL domains, the enzyme could adapt its conformation to fit the particular glycoprotein it has bound and needs to re-glucosylate.
Related publication: Roversi P, Marti L, Caputo AT, Alonzi DS, Hill JC, Dent KC, Kumar A, Levasseur MD, Lia A, Waksman T, Basu S, Soto Albrecht Y, Qian K, McIvor JP, Lipp CB, Siliqi D, Vasiljevic S, Mohammed S, Lukacik P, Walsh MA, Santino A, Zitzmann N. Interdomain conformational flexibility underpins the activity of UGGT, the eukaryotic glycoprotein secretion checkpoint. Proceedings of the National Academy of Sciences 114(32), 8544-8549, doi:10.1073/pnas.1703682114 (2017).
Diamond Light Source is the UK's national synchrotron science facility, located at the Harwell Science and Innovation Campus in Oxfordshire.
Copyright © 2022 Diamond Light Source
Diamond Light Source Ltd
Diamond House
Harwell Science & Innovation Campus
Didcot
Oxfordshire
OX11 0DE
Diamond Light Source® and the Diamond logo are registered trademarks of Diamond Light Source Ltd
Registered in England and Wales at Diamond House, Harwell Science and Innovation Campus, Didcot, Oxfordshire, OX11 0DE, United Kingdom. Company number: 4375679. VAT number: 287 461 957. Economic Operators Registration and Identification (EORI) number: GB287461957003.