Keep up to date with the latest research and developments from Diamond. Sign up for news on our scientific output, facility updates and plans for the future.
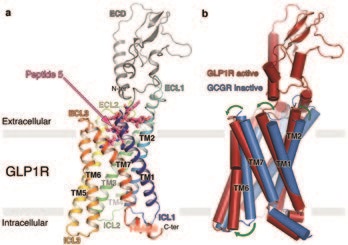
G protein-coupled receptors (GPCRs) are cell surface transmembrane receptors that transmit extracellular signals into intracellular signalling cascades. They are delicate molecular rheostats adopting multiple conformational states in response to specific hormonal and metabolic stimuli. One subfamily of GPCRs is the multi-domain class B secretin-like family, members of which are characterised by an extracellular domain (ECD) that binds peptide hormones juxtaposed to a transmembrane domain (TMD), and are validated drug targets for numerous diseases including osteoporosis, depression and anxiety and type 2 diabetes.
GPCRs’ inherent flexibility, which is integral to their function, is the cause of their extreme instability when they are extracted from their native cell membrane environment. To obtain an X-ray structure of GLP1R the receptor had to first be thermostabilised using a radiolabelled truncated and modified version of the GLP-1 agonist peptide. The final thermostabilised (StaR) GLP1R contains eleven thermostabilising amino-acid mutations, which facilitated expression and purification of the receptor and trapped it in an active conformation. Peptide 5 was designed in-house and despite being just a third of the size of native GLP-1, activates the receptor with comparable efficiency.
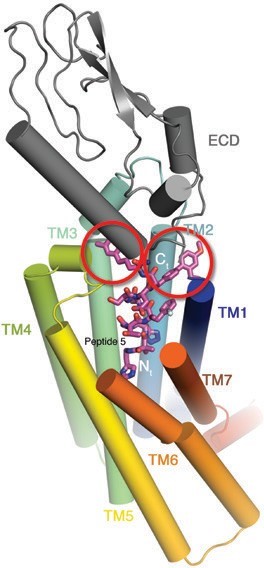
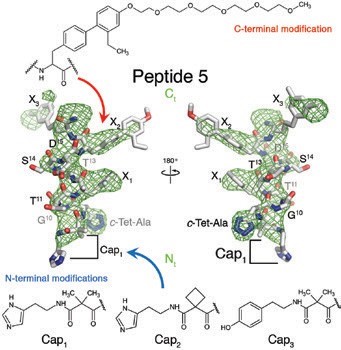
Diabetes and obesity are serious public health concerns which put great pressure on healthcare systems across the globe. Developing effective and affordable drugs to treat these conditions remains a pressing need. With the GLP1R structure in hand, the perceived historical lack of success in developing small molecules that adequately target and activate GLP1R can now be explained by the extent and complexity of interactions between hormone and receptor (Fig. 2). Guided by the structure, we have rationally designed a series of truncated peptide agonists (Fig. 3) with improved pharmacokinetic properties and important insulinotropic activity, as demonstrated through in vivo diabetic mouse models. Reducing the size of these peptides drops the manufacturing costs, making them more affordable and easier to administer. Increasing their half-life and bioavailability prolongs their activity and consequently reduces the frequency with which they need to be delivered. To conclude, this landmark structure not only aided the development of new peptide agonists against GLP1R but also concomitantly increased our understanding of the general mechanism of activation of the class B secretin group of GPCRs which includes many other important drug targets.
Jazayeri A, Rappas M, Brown AJH, Kean J, Errey JC, Robertson NJ, Fiez-Vandal C, Andrews SP, Congreve M, Bortolato A, Mason JS, Baig AH, Teobald I, Doré AS, Weir M, Cooke RM, Marshall FH. Crystal structure of the GLP-1 receptor bound to a peptide agonist. Nature 546, 254-258, doi: 10.1038/nature22800 (2017).
Diamond Light Source is the UK's national synchrotron science facility, located at the Harwell Science and Innovation Campus in Oxfordshire.
Copyright © 2022 Diamond Light Source
Diamond Light Source Ltd
Diamond House
Harwell Science & Innovation Campus
Didcot
Oxfordshire
OX11 0DE
Diamond Light Source® and the Diamond logo are registered trademarks of Diamond Light Source Ltd
Registered in England and Wales at Diamond House, Harwell Science and Innovation Campus, Didcot, Oxfordshire, OX11 0DE, United Kingdom. Company number: 4375679. VAT number: 287 461 957. Economic Operators Registration and Identification (EORI) number: GB287461957003.