Keep up to date with the latest research and developments from Diamond. Sign up for news on our scientific output, facility updates and plans for the future.
Materials that have long range magnetic order are fundamental to modern technologies, including data storage devices, space research, cancer therapy and biomedical imaging techniques. Future miniaturisation of these components depends on designing molecules that behave in the same way as larger magnets, and to do this we need an improved understanding of the connection between molecular structures and their resulting properties.
Although high-temperature- and high-magnetic-field-based studies are common in molecular chemistry, the effect of high pressures is less well explored. A team of researchers has therefore studied the effect of increasing hydrostatic pressure on rhenium(IV) compounds that exhibit long range magnetic order. They used small-molecule single-crystal diffraction on Diamond's I19 beamline to provide high-precision measurements able to monitor small changes in the structure, and high-pressure magnetometry to explore the link between the structural changes and changes in magnetic behaviour.
Their results showed that applying pressure to single crystals of rhenium(IV) compounds shortens distances between molecules in the crystal, with corresponding changes to magnetic behaviour, including an increase in the temperature at which long range magnetic order occurs.
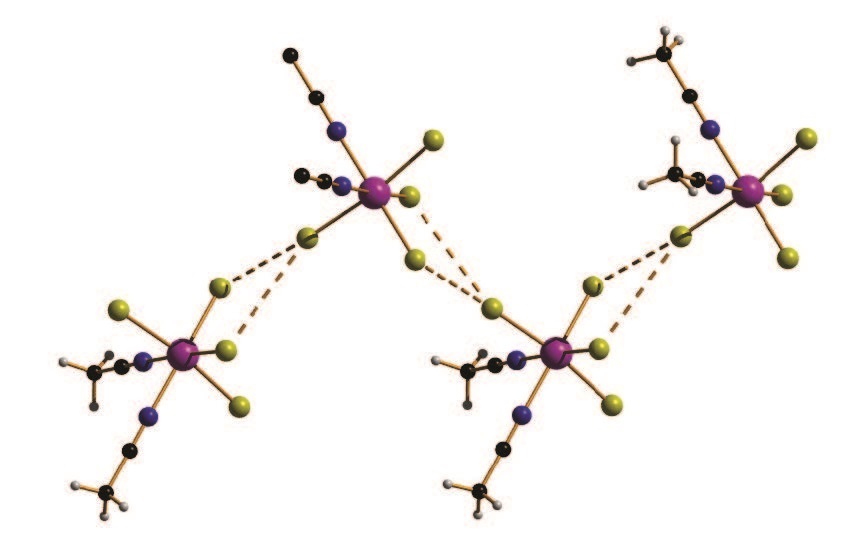
[ReCl4(MeCN)2]·MeCN (1) (Me=methyl) and [ReBr4(bpym)] (2) (bpym=2,2′-bipyrimidine) are characterised by large magnetic anisotropies and significant intermolecular magnetic exchange interactions mediated through Re–X···X–Re type contacts (X = halogen), resulting in spin canting (the non-collinearity of neighbouring spin centres) and magnetic order at low temperature (Tc (1) = 6.5 K, Tc (2) = 18.0 K)1,2,3. The dipolar exchange pathways arise as a result of the spin density from the metal ion being delocalised onto the peripheral atoms of the ligand. Intermolecular magnetic exchange interactions can be modified by changing intermolecular distances, i.e. making these distances shorter would be expected to increase the strength of the exchange and increase the ordering temperature, and one method of achieving this is to exert hydrostatic pressure. By combining high-pressure single-crystal X-ray crystallography and high-pressure SQUID magnetometry, in tandem with detailed theoretical analysis, the relationship between changing structure and changing magnetic behaviour can be extracted, and the underlying physical origins of the behaviour revealed.
Compounds 1 (Pnma) and 2 (P212121) crystallise in orthorhombic space groups, with the metal ion occupying a distorted octahedral environment, bonded to two N-atoms and four halides. The extended structures display staggered zig-zag chains that propagate along the a-axis of the unit cell via interhalide dispersive interactions (Fig. 1). Under pressures of up to 4.30 GPa unit cell volumes decrease by 20% (1) and 14% (2), the principal component of compression occurring along the c-axis of the unit cell in both cases. 2 undergoes a phase transition between 1.93 GPa and 3.06 GPa to a monoclinic phase with space group P21, and when subjected to complete compressiondecompression cycles to 4 GPa, both 1 and 2 return to their original ambient pressure unit cell/space group. In addition, ambient pressure crystal structures solved at T = 4 K show both species retain their room temperature structures. The most significant structural changes observed under pressure in 1 and 2 occur in the intermolecular Re-X···X–Re distances. For 1 the Re–Cl(4)···Cl(2)– Re distance shortens by ∼0.6 Å to 3.362(11) Å, while for 2 the Re-Br(2)···Br(4)- Re distance decreases by 0.31 Å to 3.5718(2) Å.
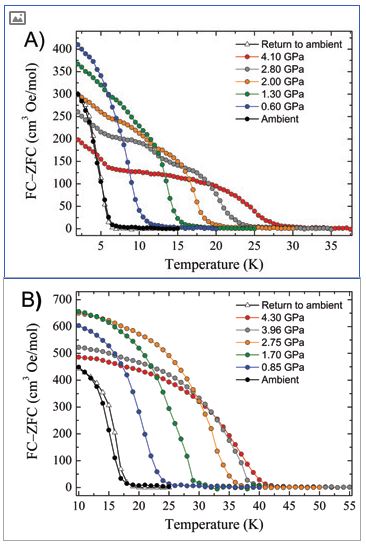
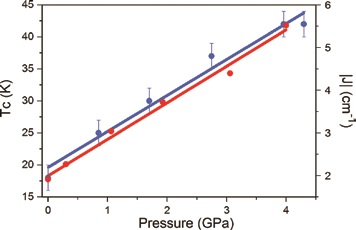
High pressure SQUID magnetometry, performed on microcrystalline samples of 1 and 2 in a turnbuckle diamond anvil cell4, shows that the application of hydrostatic pressure leads to an increase in ordering temperature (Fig. 2). At the highest pressures measured, TC (determined from field cooled and zero-field cooled measurements as a function of temperature) reaches 28 K at 4.10 GPa for 1, and 42 K at 4.30 GPa for 2. For 1, this represents a four-fold enhancement, with the rate at which TC increases being 5.1 and 5.4 K GPa−1 for 1 and 2, respectively. The phase transition observed for compound 2 does not affect the monotonic increase of the ordering temperature, and in both cases the changes in Tc are reversible; recovery of the ambient pressure TC is observed when the pressure is released from the cell. Density functional calculations show that the shortest Re-X···X-Re pathways generate the most efficient magnetic exchange interactions (J) at all pressures, and that an increase in the magnitude of J in 1 and 2 is observed when external pressure is applied, in agreement with increasing Tc in both systems. Indeed, the correlation between parameters is straightforward, external applied pressure produces a linear increase in J and a linear increase in Tc (Fig. 3).
The use of multiple high pressure techniques is a simple and efficient methodology for probing structure-property relationships. For synthetic chemists it has the added advantage of negating the need to make families of compounds that may differ electronically. In this instance, the pressureinduced enhancement of the magnetic ordering temperatures of two spin-canted ReIV systems can be directly correlated to changes in structure, specifically Re-X···X-Re distances, through the combined use of single-crystal X-ray crystallography and SQUID magnetometry, allowing unprecedented insight into the underlying physical origins of behaviour in a step-by-step manner.
Funding acknowledgement: EPSRC EP/N01331X/1 “Putting the Squeeze on Molecule-Based Magnets” We acknowledge Diamond Light Source for time on Beamline I19 under Proposal MT11879.
Corresponding author: Prof Euan K. Brechin, EaStCHEM School of Chemistry, The University of Edinburgh, ebrechin@ed.ac.uk
Woodall CH, Craig GA, Prescimone A, Misek M, Cano J, Faus J, Probert MR, Parsons S, Moggach S, Martínez-Lillo J, Murrie M, Kamenev KV, Brechin EK. Pressure induced enhancement of the magnetic ordering temperature in rhenium(IV) monomers. Nature Communications 7, 13870 doi:10.1038/ncomms13870 (2016).
Diamond Light Source is the UK's national synchrotron science facility, located at the Harwell Science and Innovation Campus in Oxfordshire.
Copyright © 2022 Diamond Light Source
Diamond Light Source Ltd
Diamond House
Harwell Science & Innovation Campus
Didcot
Oxfordshire
OX11 0DE
Diamond Light Source® and the Diamond logo are registered trademarks of Diamond Light Source Ltd
Registered in England and Wales at Diamond House, Harwell Science and Innovation Campus, Didcot, Oxfordshire, OX11 0DE, United Kingdom. Company number: 4375679. VAT number: 287 461 957. Economic Operators Registration and Identification (EORI) number: GB287461957003.