Keep up to date with the latest research and developments from Diamond. Sign up for news on our scientific output, facility updates and plans for the future.
Building on previous work conducted at Diamond, researchers have used X-ray Pair Distribution Function (XPDF) on the I15-1 beamline to probe the structure of a Metal Organic Framework (MOF) in its glassy state, from which they inferred the structure of its liquid form.
MOFs are crystalline materials, which are receiving a lot of interest due to their porous nature, which allows them to accommodate guest molecules within their structure. Potential applications include drug delivery, catalysis, and the capture or separation of gases. There are around 60,000 MOFs, and until very recently research had focused on their solid form.
For this new study, the team investigated the structure of ZIF-4 at Diamond, the Advanced Photon Source in Chicago and ISIS Neutron and Muon source, using their results to produce a computational model of the glassy state from which they could infer the structure of the liquid state. Their results show that this MOF retains its porous structure in its liquid form. This opens up a whole new area of research, investigating whether other MOFs also behave in this way, as well as discovering the properties and potential applications of liquid and glass MOFs.
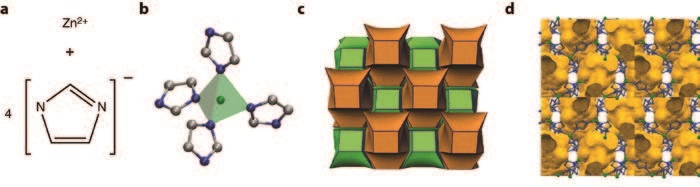
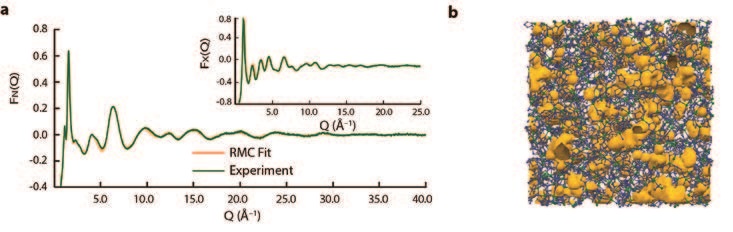
A sample of the melt-quenched glass arising from ZIF-4 was prepared ex situ, before X-ray total scattering data were collected using the I15-1 (XPDF) beamline at Diamond Light Source, UK. These data, combined with neutron data collected using the GEM Diffractometer at the ISIS Neutron and Muon Source, UK, were then modelled using reverse Monte-Carlo modelling – software which gradually alters a starting atomic configuration to fit the experimental data collected3. For this task, we used a model of an amorphous Zn(Im)2 network from a solid amorphous (non melt quenched) Zn(Im)2 phase, itself gained by adjusting a Continuous Random Network (CRN) model for aSiO2 (Fig. 2)4. The resultant configuration, the first model for a melt-quenched glass, was gained without changing network topology, and resulted in a good fit to the experimental structure factors.
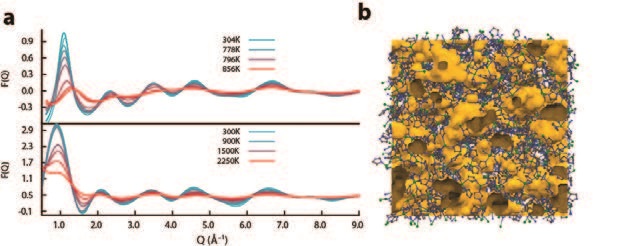
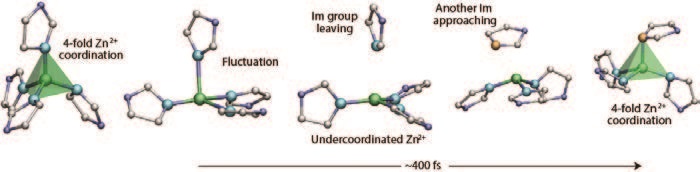
To investigate the mechanism of melting, synchrotron X-ray diffraction data were collected at the Advanced Photon Source, USA (APS), to evaluate structural changes in the glass upon heating. The pair distribution functions (PDFs) contain the expected peaks at approximately 1.3 Å, 2 Å, 3 Å, 4 Å and 6 Å, which correspond to C–C/C–N, Zn–N, Zn–C, Zn–N and Zn–Zn pair correlations, respectively. Constant temperature molecular dynamics simulations were also carried out, with the PDFs obtained showing similar trends to the experimental ones (Fig. 3). The high temperature experimental data on the liquid MOF phase at 865 K was modelled using RMC Profile3, providing the first configuration for a liquid MOF (Fig. 3b). Simulation also provided the opportunity to build up a molecular picture of the melting process (Fig. 4), showing it to proceed via dissociation of an imidazole ligand from a Zn2+ centre, followed by association of a different Im ligand – not unlike hydrogen bond switching between water molecules.
Importantly, analysis of the pore volume of the ambient temperature glass using a probe diameter of 2.4 Å showed only 4.8% accessible volume. Upon melting at 865 K, this increased to 16.2 %. Hence, our conclusion was that even at high temperatures, the liquid contains porosity. This is also greater than ionic liquids containing the imidazolium ion (C3H5N2+). Elaborating on the generality of the melting phenomenon will be important, as the activation energy required to break the coordination metal-ligand bonding is above that which causes thermal decomposition of some MOFs, including the prototypical ZIF-8.
Given the retention of coordinative bonding between crystal and liquid forms, the term ‘MOF-liquid’ was introduced, to refer to liquids formed from melting crystalline MOF structures. The research will add to the developing community of researchers interested in the combination of flexibility, defects and disorder in MOFs5.
Funding acknowledgement: We thank A. Boutin, A. Fuchs, A. Cheetham and R. Vuilleumier for fruitful discussions. This work benefited from the financial support of ANRT (thèse CIFRE 2015/0268). We acknowledge access to HPC platforms provided by a GENCI grant (A0010807069). T.D.B. would like to thank the Royal Society for a University Research Fellowship.We also thank Diamond Light Source for access to beamline I15-1 (EE15676), and D. Keeble and P. Chater for assistance with data collection on I15-1 during its initial commissioning phase. We gratefully acknowledge the Science and Technology Facilities Council (STFC) for access to neutron beamtime at ISIS on the GEM instrument. This research used resources of the Advanced Photon Source (Beamline 11-ID-B, GUP44665), a US Department of Energy (DOE) Science User Facility operated for the DOE.
Corresponding author: Dr Thomas Douglas Bennett, University of Cambridge, tdb35@cam.ac.uk
Gaillac R, Pullumbi P, Beyer KA, Chapman KW, Keen DA, Bennett TD, Coudert F-X. Liquid metal–organic frameworks. Nature Materials 16, 1149-1154, doi:10.1038/nmat4998 (2017).
Diamond Light Source is the UK's national synchrotron science facility, located at the Harwell Science and Innovation Campus in Oxfordshire.
Copyright © 2022 Diamond Light Source
Diamond Light Source Ltd
Diamond House
Harwell Science & Innovation Campus
Didcot
Oxfordshire
OX11 0DE
Diamond Light Source® and the Diamond logo are registered trademarks of Diamond Light Source Ltd
Registered in England and Wales at Diamond House, Harwell Science and Innovation Campus, Didcot, Oxfordshire, OX11 0DE, United Kingdom. Company number: 4375679. VAT number: 287 461 957. Economic Operators Registration and Identification (EORI) number: GB287461957003.