Related publication:
Bennett TD, Tan J-C, Yue Y, Baxter E, Ducati C, Terrill NJ, Yeung HHM, Zhou Z, Chen W, Henke S, Cheethan AK, Greaves GN. Hybrid glasses from strong and fragile metal-organic framework liquids. Nature Communications 6, doi:10.1038/ncomms9079 (2015).
Keywords:
Chemical sciences; Inorganic chemistry; Materials science.
Hybrid glasses are a new variety of glass that have been discovered and reported in Nature Communications, assisted by in situ X-ray scattering experiments on the Small Angle Scattering and Diffraction beamline (I22). These new glasses comprise inorganic metals, like zinc, interlinked by organic bridges, such as immidazolate ligands. They have been obtained starting from metal organic frameworks (MOFs), an innovative range of novel micro-engineered crystalline materials. MOFs have attracted much excitement recently on account of their wide-ranging potential as materials for gas separation and storage, biomedical and pharmaceutical applications, as well as smart sensors and microelectronic systems. It is too early to say where the new hybrid glasses will offer most potential, but the fact that they can be cast, means that fabrication into different sizes and shapes will be possible, otherwise precluded using crystalline powders. Although the glasses most familiar to the public are based on silicates and have their origins over 5000 years ago, many new families of glass have been developed in the last 50 years: those incorporating chalcogenides; glasses containing halogens; metallic glasses; molecular glasses like water, and so on. This huge diversity demonstrates how the glassy state has become the alternative to the crystalline state, both forms of solid state being obtained by cooling from the liquid state, processes often most dramatically portrayed in steel plants and in the glass industry. In addition, new ways of glass formation have evolved in the laboratory, including amorphisation, whereby crystalline materials are transformed into amorphous solids by applying modest pressure or temperatures well below the melting point Tm. The inspiration for the present work was to reveal a completely new variety of glass based on organic-inorganic systems, and to create versions both by amorphisation and also by melt-quenching. These processes have been elucidated using I22, which, with advanced differential scanning calorimetry (DSC), proved critical in following the dynamic transformation from crystalline precursor, to stable hybrid glass, as well as the formation of hybrid liquids and glasses melt-quenched from them.
Soft Condensed Matter Village | Beamline I22
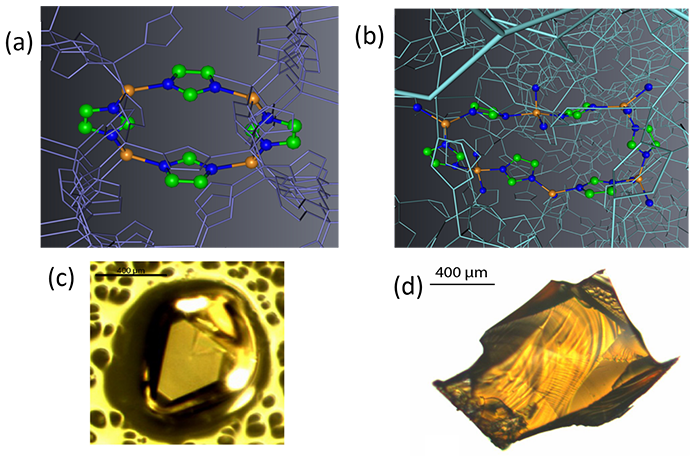
Figure 1: Visualisation of the structure of crystalline ZIF-4 (a) and amorphised ZIF-4 (b), highlighting a 6-fold ring. Optical Images of crystalline (c) and hybrid melt-quenched glass (d).
Metal organic frameworks, a new class of novel crystalline materials, show exceptional promise in areas as diverse as carbon storage, drug sequestration and water desalination1. They contain an important subgroup of 3-dimensional networks, zeolitic immidazolate frameworks (ZIFs). Many of these have structures equivalent to inorganic zeolites, except that tetrahedral silicon and aluminium are replaced by metal cations and bridging oxygens by immidazolate linkages (Fig. 1). In earlier work on zeolites at the Daresbury Synchrotron Radiation Source, it was explained how these inorganic frameworks collapse through amorphisation to glasses of very similar density and structure to melt-quenched glasses2. The transformation appears to be driven by the coexistence of a low density liquid (LDL) of low fragility and a high density liquid (HDL) of high fragility, at temperatures close to the respective glass transitions Tg of the corresponding glasses (LDA and HDA)2. LDA phases have the attributes of perfect glasses3 – similar in entropy to the crystalline phase but aperiodic. For this work, in situ Small Angle Scattering (SAXS) proved to be a key technique2.
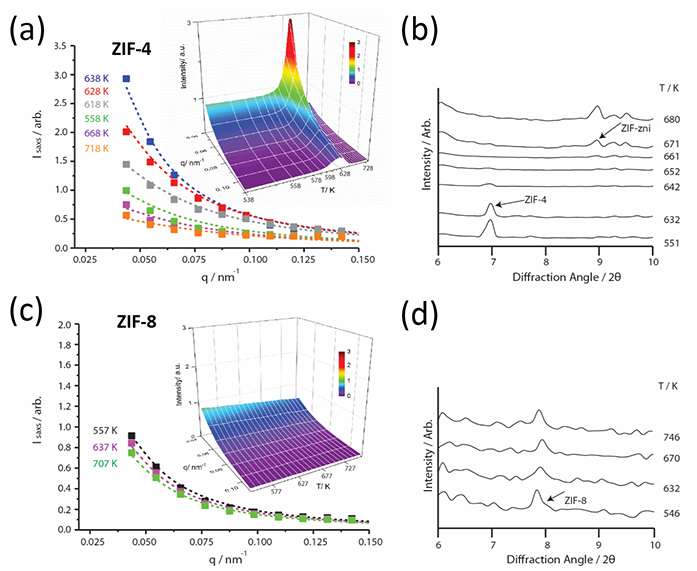
Figure 2: In situ temperature dependence of SAXS and XRD for ZIF-4 (a,b) and ZIF-8 (c,d) measured on I22. Curves are Lorentzian fits to SAXS profiles at selected temperatures. Hybrid HDA glass forms at the end of the ZIF-4 peak but ZIF-8 remains crystalline.
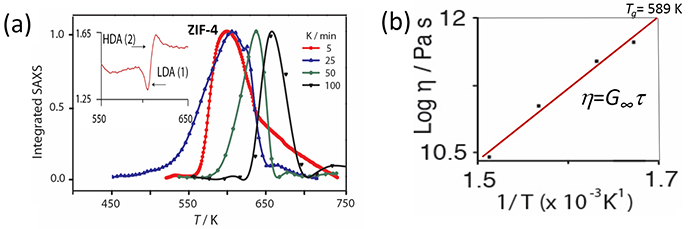
Figure 3: Obtaining from in situ SAXS the viscosity of the LDA hybrid glass controlling the collapse of ZIF-4. a) Integrated SAXS covering the collapse of ZIF-4 (inset DSC amorphisation signature); b) Kinetic viscosity h and glass transition Tg for initial LDL phase.
Furthermore, where substantial increases in the amorphisation temperature TA with the rate of zeolite collapse had previously been found2,3, the same behaviour was also discovered for ZIF-4, the peak in the integrated SAXS rising in TA by almost 100 degrees when the heating rate increased by a factor of 20 (Fig. 3a). The rate of collapse was taken to be governed by the kinetic viscosity of the LDL phase2, which can be obtained from the Maxwell relation h=G∞t. Here G∞ is the instantaneous shear modulus of ZIF-4 and t is the reciprocal of the heating rate achieved with the Linkam furnace (Fig. 3b). The glass transition temperature of the LDA perfect glass T(LDA)g is then given by the temperature where h=1012 Pas (Fig. 3b). From the slope of Angell plot log h=Tg /T at T=Tg , the LDL melt fragility m can be obtained (Fig. 4a). As occurs with zeolites3, m=14, a fragility much lower than silica, for example, identifying ZIF-4 (LDL) as a superstrong liquid. Very similar values have obtained from DSC, all of which demonstrates how SAXS on I22 can be used to determine Tg and m for liquids during amorphisation. In sharp contrast, the fragility of the final HDL phase is much larger m=41. The viscous dynamics of these two hybrid liquids are contrasted in Fig. 4a, compared to inorganic oxides like molten silica and anorthite.
An important empirical relation Tg=2/3Tm relates the glass transition and melting temperatures of standard glass-forming liquids3. Originally established for molecular liquids, this relationship has been extended in the Nature Communications article to include oxides, now covering melting temperatures of more than 2000 degrees (Fig. 4b). For ZIF-4 Tg projects a melting temperature Tm , 50 degrees below thermal decomposition and in close agreement with the DSC melting signature defining Tm. Cooling this hybrid liquid results in a hybrid melt-quenched glass (Fig. 1d), whose structure factor is indistinguishable from that of the hybrid HDA glass formed by amorphisation. This coincidence points to the two hybrid glasses occupying similar minima in the potential energy landscape.
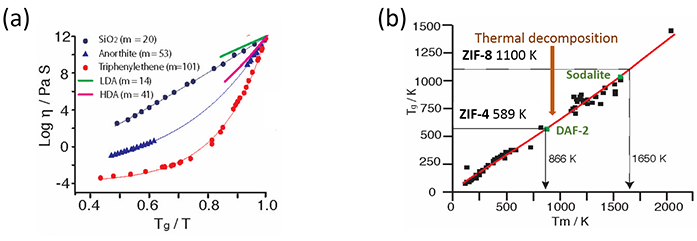
Figure 4: Fragilities of the polyamorhic LDL and HDL hybrid phases of ZIF-4, the glass transition Tg where collapse occurs, the ultimate melting point Tm, and the decomposition temperature. The equivalent virtual Tg and Tm values extrapolated for ZIF-8 from pressure amorphisation metrics both of which occur beyond decomposition. a) Low and high fragilities of hybrid LDL and HDL phases, compared to standard glass formers; b) Empirical Tg=2/3Tm Law and projection of Tms from Tgs for ZIF-4 and ZIF-8, compared to inorganic zeolites DAF-2 and silicalite, respectively.
ZIF-8, on the other hand, shows no such melting feature, decomposing before it amorphises or even melts (Fig. 2c). This work has shown how a hypothetical Tg can be estimated from the pressure of amorphisation PA , which is known to take place at room temperature. This Tg is far greater than that of ZIF-4 and of the decomposition temperature shared by both. The Tg of ZIF- 8 projects a hypothetical melting temperature falling among the inorganic minerals (Fig. 4b). In particular, both the Tms of ZIF-4 and ZIF-8 lie very close to those of zeolites DAF-2 and sodalite respectively. The exciting conclusion is that melting of framework materials may well be related to network topology and collective vibrations, rather than nearest neighbour geometry and stretching oscillations - the tenet of the well-known Lindemann Law which asserts that melting is driven by vibrations of nearest neighbour atoms. The research, fuelled by experiments on I22, has led to the discovery of a completely new class of glasses: those based on organic-inorganic or hybrid structures. As part of this research the evolution of hybrid glass formation, from amorphisation to melt-quenching, has been elucidated, as well as the prediction of the virtual temperature at which some hybrid materials would melt, if they did not decompose first.
References:
- Furukawa, H., Cordova, K. E., O’Keeffe, M. & Yaghi, O. M. The chemistry and applications of metal-organic frameworks. Science 341, 974-986 (2013), doi:10.1126/science.1230444 (2013).
- Greaves, G. N. et al. The rheology of collapsing zeolites amorphized by temperature and pressure. Nature Materials 2, 622-629, doi:10.1038/ nmat963 (2003).
- Greaves, G. N. & Sen, S. Inorganic glasses, glass-forming liquids and amorphizing solids. Advances in Physics 56, 1-166, doi:10.1080/00018730601147426 (2007).
Funding Acknowledgements:
The work was principally enabled by the award of Diamond beamtime on I22 (SM5692). The authors would also like to thank Trinity Hall (T.D.B.); Sidney Sussex College (GNG); Wuhan University of Science and Technology (Y.Z.Y. and G.N.G.), ERC grant number 259619 PHOTO EM (C.D.); and the Alexander von Humboldt Foundation (S.H.)
Corresponding Author:
Professor Neville Greaves, Aberystwyth University, [email protected].


 Diamond Annual Review 2016
Diamond Annual Review 2016

