Summary
In light of the ongoing coronavirus (COVID-19) pandemic, the Ivan Ahel Laboratory at the University of Oxford joined together with the XChem team at Diamond Light Source to contribute to the current global efforts discovering and developing much-needed novel antiviral therapeutic possibilities.
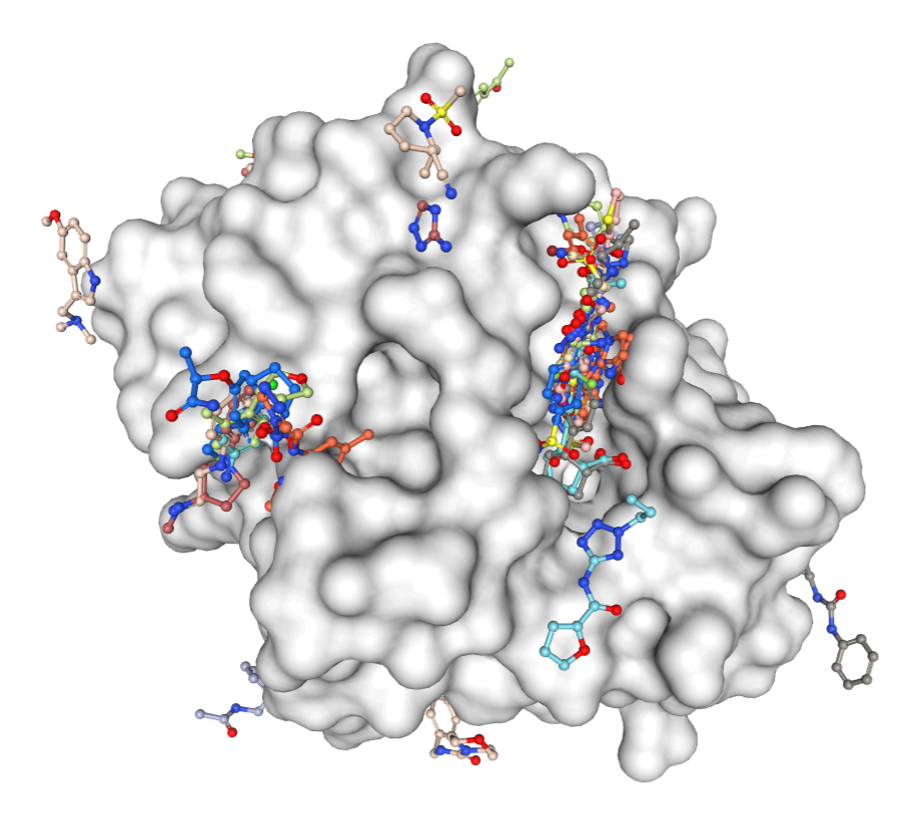
For this, the Ivan Ahel team has been able to rapidly solve the SARS-CoV-2 macrodomain structure to near atomic resolution, and then, together with Frank von Delft’s XChem team, completed a large crystallographic fragment screen against this target.
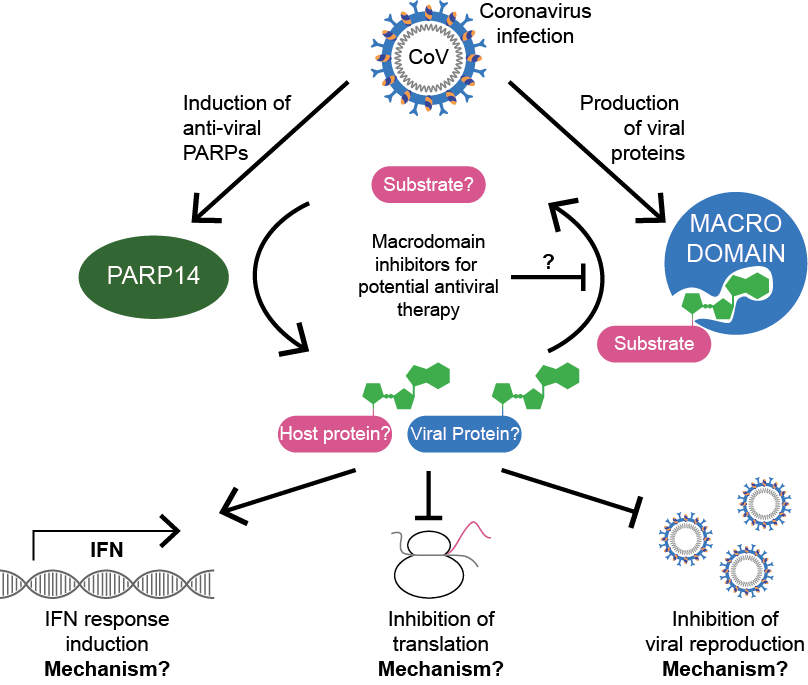
The macrodomain (also called Macro X domain) is a 150 amino acid protein module with (ADP-ribosyl)-hydrolase activity that is a part of the SARS-CoV-2 multidomain protein nsp3. ADP-ribosylation is a reversible post-translational modification of proteins synthesised by the PARP family of enzymes, and regulates many pathways in human cells, including the DNA damage response and antiviral defences. Several of the human PARPs such as PARP10, PARP13 and PARP14 act specifically as antiviral proteins to generate an antiviral environment and prevent virus replication. In contrast, the viral macrodomain removes the ADP-ribosylation modifications, thereby enabling the virus to counteract the PARP-induced innate immunity (Fehr et al, Trends Microbiol, 2018; Figure 1).
The macrodomain enzyme represents a promising drug target for the treatment of coronavirus infections, since macrodomain-deficient viruses (including all studied coronaviruses, alphaviruses and hepatitis E virus) are unable to replicate in human cells (eg. Fehr et al, mBio, 2016). Nevertheless, no inhibitors for this enzyme have been developed to date.
Figure 1. Model for the role of ADP-ribosylation following coronavirus infection. Infection of cells with coronavirus leads to the accumulation of human antiviral proteins as well as viral proteins, such as human PARP14 and viral macrodomain, respectively. The ADP-ribosylation activity of PARP14 leads to the induction of the interferon (IFN) response, down-regulation of translation and prevents viral replication. The viral macrodomain exhibits an (ADP-ribosyl)-hydrolase activity which is required for evasion of immune responses and efficient viral replication through antagonizing PARP14 activity.
Diamond Light Source is the UK's national synchrotron science facility, located at the Harwell Science and Innovation Campus in Oxfordshire.
Copyright © 2022 Diamond Light Source
Diamond Light Source Ltd
Diamond House
Harwell Science & Innovation Campus
Didcot
Oxfordshire
OX11 0DE
Diamond Light Source® and the Diamond logo are registered trademarks of Diamond Light Source Ltd
Registered in England and Wales at Diamond House, Harwell Science and Innovation Campus, Didcot, Oxfordshire, OX11 0DE, United Kingdom. Company number: 4375679. VAT number: 287 461 957. Economic Operators Registration and Identification (EORI) number: GB287461957003.