____________________________________
Industrial Liaison Group:
Tel: +44 (0) 1235 778797
E-mail: [email protected]
Elastin allows tissues in humans and other mammals to stretch and return to original shape e.g. during respiration or heart beats. The schematic on the right shows how many tropoelastin monomers (blue) can selfassemble and cross-link (red) to form elastin but the structure of the soluble precursor of elastin, tropoelastin, is not well understood.
Due to its flexible nature and strong tendency to self-associate, tropoelastin is not a good candidate for crystallographic studies. The team needed an alternative method of investigating the structure of a flexible protein in solution.
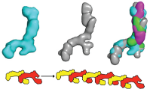
An international team from UK, Australia, USA and Europe used beamline I22 at Diamond (along with ESRF and APS) to investigate the 3D solution structure of tropoelastin using small angle X-ray scattering. Complementary small angle X-ray (left) and neutron scattering (centre) measurements allowed identification of discrete regions of the molecule. The combined data provided details of the full length tropoelastin structure (right).
The small angle scattering measurements allowed the researchers to gain high quality structural information from a flexible protein. They gained an understanding of the subunit structure that governs the ability of elastin to stretch and how it attaches to cells. The team were able use this information to propose a mechanism for the self-assembly of tropoelastin to form elastin; nature’s ideal nanoscale spring.



“We discovered that tropoelastin is a curved, spring-like molecule with a ‘foot’ region to facilitate attachment to cells.”
Dr Clair Baldock, Wellcome Trust Centre for Cell Matrix Research, The University of Manchester

Diamond Light Source is the UK's national synchrotron science facility, located at the Harwell Science and Innovation Campus in Oxfordshire.
Copyright © 2022 Diamond Light Source
Diamond Light Source Ltd
Diamond House
Harwell Science & Innovation Campus
Didcot
Oxfordshire
OX11 0DE
Diamond Light Source® and the Diamond logo are registered trademarks of Diamond Light Source Ltd
Registered in England and Wales at Diamond House, Harwell Science and Innovation Campus, Didcot, Oxfordshire, OX11 0DE, United Kingdom. Company number: 4375679. VAT number: 287 461 957. Economic Operators Registration and Identification (EORI) number: GB287461957003.