In October 2023, the Nobel Prize in Chemistry was awarded to three scientists (Aleksey Yekimov, Louis Brus and Moungi Bawendi) for their roles in the discovery and development of quantum dots (QDs). At the nanoscale, the properties of matter are determined by quantum phenomena, and governed by size. QDs are nanoparticles of light-emitting semiconductors, so tiny that the wavelength of light they emit is determined by their size, not their chemistry. By carefully controlling the synthesis process, manufacturers can create QDs that emit light across the breadth of the visible spectrum and into the infrared. QDs are already used for QLED televisions and computer monitors and for medical applications such as identifying and mapping tumours. However, most are based on cadmium selenide, a toxic metal system that creates challenges for manufacturing, disposal and medical use. Indium phosphide (InP), already in commercial use, is a promising replacement. However, the standard synthesis of InP requires the use of tris(trimethylsilyl)phosphine, a highly reactive and dangerous phosphorous precursor. In work recently published in Nanoscale Horizons, researchers from King's College London, the University of Oxford, the University of Cambridge, London South Bank University and Indiana University developed a significantly simpler synthetic methodology using a solid, air- and moisture-tolerant primary phosphine. This new methodology greatly improves the ease of QD synthesis in the lab, and potentially in industry.
Prof Mark Green, from King's College London introduces the significance of quantum dots;
This year's Nobel Prize for Chemistry has really propelled quantum dots into the public consciousness. They're exciting because they allow us to take a semiconductor that isn't normally tuneable and tune it so that it emits particular wavelengths of light. And we do that by carefully controlling the size of the particles we create. Quantum dots are already in use commercially, in TVs, and for biological labelling - making tumours glow, for example.
Although physicist Herbert Fröhlich predicted that nanoparticles would have unusual characteristics in 1937, it wasn't until the 1970s that technology evolved far enough to make them. Even then, they were challenging to produce, and few people considered that these fascinating novelties could be put to practical use.
Aleksey Yekimov succeeded in creating size-dependent quantum effects in coloured glass in the early 1980s, swiftly followed by Louis Brus' discovery of size-dependent quantum effects in particles floating freely in a solution.
However, producing consistent, high-quality quantum dots remained a problem until 1993. Then Moungi Bawendi and his research group at the Massachusetts Institute of Technology, MIT, developed an easy way to grow nanocrystals of a specific size by dynamically varying the temperature of the solution. Researchers could finally explore the full potential of QDs.
The beauty of Bawendi's synthesis method is that it allows you to control exactly how these particles grow
Prof Green says,
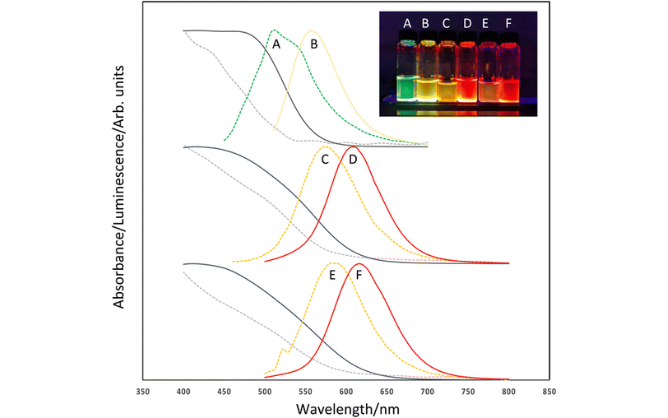
They grow slowly enough that you can follow them using lab spectroscopy, take samples out of your flask and isolate the individual fractions. From one reaction, you can produce QDs all the way through from blue to red. The problem is that the chemistry is based on cadmium, and the metal's toxicity causes problems during manufacturing and disposal and is a concern for medical applications.
The leading candidate for cadmium-free QDs is indium phosphide (InP), which is already used commercially for QLED televisions. However, the production of InP QDs brings its own challenges. The standard synthesis model uses a volatile and highly reactive compound - tris(trimethylsilyl)phosphine - as a phosphorous precursor.
Prof Green explains;
What we wanted to do was come up with a really simple, green way to make InP QDs. That meant finding a better phosphorus precursor, and we have developed a new synthesis route based on phosphinecarboxamide, which is a stable, crystalline solid that's much easier to handle.
And it seems as though the team has done just that. However, when you get down to the quantum scale, things get more complicated. The surface of the material becomes all-important, so any defects or oxidation can affect the optical properties. The performance of the QDs is improved by adding zinc (which enhances their optical brightness) and coating them with zinc sulfide, to give them a core/shell architecture.
Prof Green continues;
We were aiming for a InZnP/ZnS core/shell structure, but how do we know we've achieved that, rather than simply a big mix of indium, zinc, phosphorus and sulfur? We brought our QDs to the electron Physical Science Imaging Centre (ePSIC), which is one of the few facilities where we can look at these particles under a microscope. We can see the particles, and we can do an elemental analysis to see which elements are there, and where they are. We can check we've got the crystalline structure. And without looking at the particles, we can't know we've made what we hope we've made, so ePSIC is vital to this research.
The team need to do more analysis, but their initial results suggest that they have done exactly what they set out to do - make luminescent QDs using a safe phosphorus source. That means - for QDs at least - the future could be very bright indeed.
To find out more about the electron Physical Science Imaging Centre (ePSIC) or discuss potential applications, please contact Principal Electron Microscopist Chris Allen: [email protected].
Wang Y et al. Phosphinecarboxamide based InZnP QDs–an air tolerant route to luminescent III–V semiconductors. Nanoscale Horizons (2023). DOI:10.1039/D3NH00162H.
Diamond Light Source is the UK's national synchrotron science facility, located at the Harwell Science and Innovation Campus in Oxfordshire.
Copyright © 2022 Diamond Light Source
Diamond Light Source Ltd
Diamond House
Harwell Science & Innovation Campus
Didcot
Oxfordshire
OX11 0DE
Diamond Light Source® and the Diamond logo are registered trademarks of Diamond Light Source Ltd
Registered in England and Wales at Diamond House, Harwell Science and Innovation Campus, Didcot, Oxfordshire, OX11 0DE, United Kingdom. Company number: 4375679. VAT number: 287 461 957. Economic Operators Registration and Identification (EORI) number: GB287461957003.