Aluminium hydroxide, Al(OH)3, is a lamellar material with octahedral vacancies in its layers. It has long been known that it can ‘imbibe’ LiX salts to form layered double hydroxides (LDHs) of the form [LiAl2(OH)6]X·H2O, where X is a generic anion. LDHs are important ion-exchangers, with applications in catalysis, biomedicine, and polymer science. Although size considerations should not be problematic, the incorporation of other metal ions into the octahedral vacancies in Al(OH)3 is challenging, and was not reported until 2004 when ZnII, CuII, NiII and CoII nitrates were successfully intercalated to give novel [MAl4(OH)12](NO3)2·yH2O LDHs.1 In this work, we report recent developments in the intercalation chemistry of Al(OH)3. These include a detailed structural study, the synthesis of new LDHs containing mixtures of MII cations, and a comprehensive in situ study of their intercalation reactions. Through use of the high-quality energy-dispersive diffraction capabilities of beamline I12, we were able to continuously record diffraction patterns in real time as reactions proceeded, and from these data deduce information on the reaction mechanisms. These results are crucially important in our quest to understand more about how solid state and heterogeneous solid/liquid reactions proceed.
Layered double hydroxides are a widely studied class of materials comprising positively charged layers, with charge-balancing anions located in the interlayer space. They have the generic formula [MII1-qMIIIq(OH)2]q+Xn–q/n·yH2O. LDHs have a range of potential applications, including in catalysis, biomedicine, and polymer science.2,3 They display a diverse ion-exchange intercalation chemistry, in which the initial interlayer anion is replaced by another. Generally, LDHs contain MII and MIII metal ions, with the MII:MIII ratio in the range 2:1 – 4:1. The LDH [LiAl2(OH)6]X·H2O (X = Cl–, Br–, NO3–, 0.5SO42–, etc) is prepared through the reaction of LiX salts with Al(OH)3 at around 90 °C, and is the only LDH containing mono- and trivalent metal cations. Although Li+ is similar in size to Zn2+, Cu2+, Ni2+, and Co2+, it is much harder to intercalate the latter into Al(OH)3. Successful reaction could only be achieved by first grinding γ-Al(OH)3 (gibbsite) in a ball mill, and then subjecting it to an extended hydrothermal treatment.1 The LDHs thereby generated are highly novel synthetic materials with formula [MAl4(OH)12](NO3)2·yH2O (M = Zn, Cu, Ni, Co; MAl4-NO3).
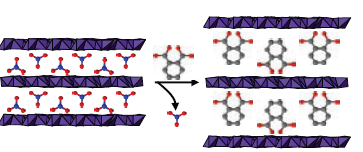 |
| Figure 1: A schematic representation of intercalation into the LDH [CoZnAl8(OH)24](NO3)4·yH2O, showing the initial nitrate anion (left) being replaced with phthalate to give [CoZnAl8(OH)24](C8H4O4)2·yH2O (right). |
The study of reaction mechanisms and kinetics has been neglected for the solid state, largely because there are few non-invasive probes available. A powerful non-invasive probe is in-situ energy-dispersive X-ray diffraction (EDXRD). The exceptional EDXRD capabilities of beamline I12 provide an unparalleled opportunity to extend our knowledge in this area, and the present study forms part of work to explore the nanoscopic processes involved in a range of solid state reaction processes.
In this study,4 we sought to expand our earlier work in a number of ways. First, we used Rietveld refinement methods to propose a structure for the new MAl4-NO3 materials. It was found that MII cations occupy half the octahedral vacancies in the Al(OH)3 layers, with almost complete ordering of MII cations. Next, we explored the possibility of incorporating salts other than nitrates, and of further reacting the MAl4-NO3 materials to fill the remaining octahedral holes. Beyond this, experiments were undertaken to prepare three-metal LDHs by reacting Al(OH)3 with mixtures of MII nitrates. The idealised formula of these materials is [MM’Al8(OH)24](NO3)4·yH2O. However, it was found that Al(OH)3 exhibits selective intercalation chemistry, imbibing metal cations preferentially in the order Li+ >> Ni2+ >> Co2+ ≈ Zn2+. By varying the ratio of metals in the reaction gel, it proved possible to control the stoichiometry of the LDHs produced. For instance, a 1 : 1 mixture of Zn and Co nitrates yields the LDH [CoZnAl8(OH)24](NO3)4·yH2O.
In order to prove definitively that the new materials prepared are layered double hydroxides, it was necessary to demonstrate that the interlayer nitrate anion could be replaced with other anions, and hence reactions were undertaken using a range of simple organic anions (see Fig. 1). It proved facile to replace the nitrate anions with a range of organic species such as phthalate (C8H4O42–).
These experiments proved that the new materials were indeed LDHs. As they are highly crystalline, with distinct and non-overlapping sets of Bragg reflections for each phase, they were chosen as ideal candidates for the first time-resolved study of a chemical reaction system to be undertaken at Diamond Light Source. The existing Oxford-Daresbury in situ reaction cell was refurbished, commissioned onto beamline I12, and detailed alignment with the I12 optics undertaken. Photographic and schematic representations of the reaction cell are given in Fig. 2.
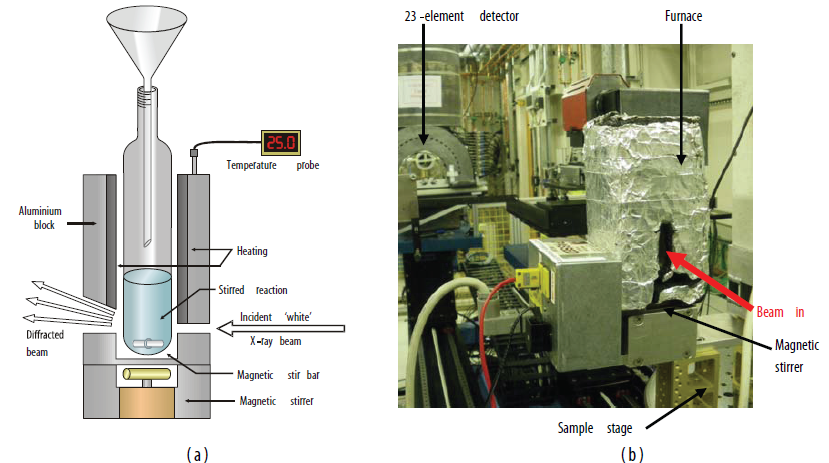 |
| Figure 2: (a) Schematic and (b) photographic representations of the in situ chemical reaction monitoring cell used on beamline I12. |
We were pleased that, with very little optimisation, it proved possible to record high-quality diffraction patterns in as little as 5 – 10 s. Unfortunately, the LDH intercalation reactions were observed to be very rapid (< 30s); hence, a syringe pump was used to add a solution of the guest to a suspension of the LDH dropwise. In order to extract all the desired chemical information, it proved necessary to move the 23-element detector system on I12 to an offset position. This required removal of a collimator, introducing extra noise into the diffraction patterns. Nevertheless, very high quality data were obtained. Data for the reaction between [CoZnAl8(OH)24](NO3)4·yH2O (CoZnAl8-NO3) and phthalate to give CoZnAl8-phthalate are shown in Fig. 3. A plot showing the variation in diffracted intensity with time is depicted in Fig. 3a. This contains only two phases: CoZnAl8-NO3 and CoZnAl8-phthalate. No crystalline intermediates may be observed.
  |
| Figure 3: EDXRD data for the conversion of CoZnAl8-NO3 to CoZnAl8-phthalate. (left) 3D stacked plot and (right) α vs. time plots showing the starting material (black) and product (red). Reproduced with permission from ref 2. Copyright Royal Society of Chemistry, 2011. |
The observed Bragg reflections were integrated and converted into the extent of reaction, α, where α = Ihkl(t) / Ihkl(max) [Ihkl is the integrated intensity of the hkl reflection]. The extent of reaction vs. time plots given in Fig. 3b show that the host and product curves cross very close to α = 0.5, suggesting that loss in diffracted intensity from the host is exactly matched by the increase in intensity from the product, and confirming a one-step I12reaction process (if intermediates were involved, then the curves would cross at α ca. 0). This is in agreement with previously reported results for the MAl4-NO3 LDHs.1
As a result of these preliminary experiments, we have now developed an optimised set-up for the study of chemical reactions on I12, and are actively working with Diamond Light Source to develop a new furnace with improved capabilities, as well as to reduce the noise introduced to the system when the detectors are moved to an offset position to access the high d-spacing region. This research forms part of a programme of investigations seeking to elucidate more detail on the nanoscopic processes involved in solid state reaction mechanisms, and the outstanding capabilities of I12 will be profoundly important in allowing us to drive this work forward.
Williams, G.R., Moorhouse, S.J., Prior, T.J., Fogg, A.M., Rees, N.H. & O’Hare, D. New insights into the intercalation chemistry of Al(OH)3. Dalton Trans. 40(22):6012-22 (2011)
AMF thanks the Royal Society for a University Research Fellowship, and SJM the EPSRC and Diamond Light Source for provision of a research studentship.
Diamond Light Source is the UK's national synchrotron science facility, located at the Harwell Science and Innovation Campus in Oxfordshire.
Copyright © 2022 Diamond Light Source
Diamond Light Source Ltd
Diamond House
Harwell Science & Innovation Campus
Didcot
Oxfordshire
OX11 0DE
Diamond Light Source® and the Diamond logo are registered trademarks of Diamond Light Source Ltd
Registered in England and Wales at Diamond House, Harwell Science and Innovation Campus, Didcot, Oxfordshire, OX11 0DE, United Kingdom. Company number: 4375679. VAT number: 287 461 957. Economic Operators Registration and Identification (EORI) number: GB287461957003.