In general, the antibiotics that are prescribed by your GP work. Complete the prescribed course and your symptoms have gone. But in some cases, the bacteria causing the illness have developed a resistance to the drugs. We need to find a way to improve these drugs so that they can overcome this resistance. But we need to know how our drugs work before we can improve them, and one way to do this is by producing a 3D molecular image of the drug/inhibitor in action. Knowing the 3D structure of an antibiotic bound to its target protein elucidates the molecular mechanism – revealing how the drug works and how it could be modified, for example, to overcome resistance.
%20sm.2020-02-21-14-24-40.png) A collaboration of scientists have used Diamond Light Source and the European Synchrotron Radiation Facility (ESRF) to solve the 3D structure of the penicillin binding protein PBP3 from Pseudomona aeruginosa (P. aeruginosa), a common bacterium which can cause disease in animals and humans. The organism is particularly dangerous to those with a compromised immune system, often causing secondary infections that can be fatal to the patient. And to make matters worse, it can easily thrive on most surfaces, including medical equipment, which can result in cross-infections in hospitals and clinics.
A collaboration of scientists have used Diamond Light Source and the European Synchrotron Radiation Facility (ESRF) to solve the 3D structure of the penicillin binding protein PBP3 from Pseudomona aeruginosa (P. aeruginosa), a common bacterium which can cause disease in animals and humans. The organism is particularly dangerous to those with a compromised immune system, often causing secondary infections that can be fatal to the patient. And to make matters worse, it can easily thrive on most surfaces, including medical equipment, which can result in cross-infections in hospitals and clinics.
The research team from the University of Oxford’s Division of Structural Biology, the Oxford Protein Production Facility (based at the Research Complex at Harwell) and the University of Dundee’s College of Life Sciences, are targeting P. aeruginosa because of its clinical prevalence and its ability to cause secondary ailments, and have selected PBP3 because it is a proven therapeutic target. The team used Diamond’s protein crystallography facilities to solve three structures: the protein in native form; and the protein linked to two important antibiotics, carbenicillin and ceftazidime.
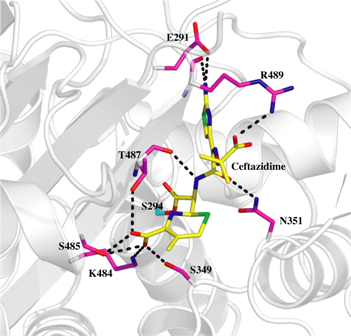
The picture to the right shows the overall structure of PBP3 with ceftazidime (yellow spheres) bound in the active site.
“The aim of the project is to understand the interaction between the antibiotic and its target at the molecular level, with a view to develop the next generation of antibiotics and combat the problem of drug resistance.”Dr Ray Owens, Oxford Protein Production Facility
Their structures suggest that there could be scope to develop new drugs that work in combination with existing PBP inhibitors to make them more effective and able to overcome resistance.
“The result is a beautiful 3D picture of how drugs work against bacteria and we can actually see the molecular basis of inhibition. Because we know how it works, we can look at the structure and see ways the drugs could be modified.”Professor William Hunter, University of Dundee
Crystal Structures of Penicillin-Binding Protein 3 from Pseudomonas aeruginosa: Comparison of Native and Antibiotic-Bound Forms
Sainsbury S, Bird L, Rao V, Shepherd SM, Stuart DI, Hunter WN, Owens RJ, Ren J.
Journal of Molecular Biology, Volume 405, Issue 1, 7 January 2011, Pages 173-184.
http://dx.doi.org/10.1016/j.jmb.2010.10.024
The Research Complex at Harwell is a multidisciplinary laboratory that provides state-of-the-art facilities for scientists to undertake cutting edge research in the life and physical sciences, and the interface between them. It is located on the Rutherford Appleton Laboratory site on the Harwell Oxford Campus, adjacent to Diamond, the new third generation Synchrotron Radiation source. It is also close to other leading facilities on the campus: the ISIS neutron source, Central Laser Facility, Membrane Protein Laboratory, MRC Mammalian Genetics Unit and Mary Lyon Centre, and a Biological Solid State NMR Facility. Access is granted, on a competitive basis, to the best research teams from UK universities, Research Councils and Diamond. The MRC is leading the project on behalf of RCUK, in partnership with BBSRC, EPSRC, NERC, STFC and Diamond. More information can be found at www.rc-harwell.ac.uk
Diamond Light Source is the UK's national synchrotron science facility, located at the Harwell Science and Innovation Campus in Oxfordshire.
Copyright © 2022 Diamond Light Source
Diamond Light Source Ltd
Diamond House
Harwell Science & Innovation Campus
Didcot
Oxfordshire
OX11 0DE
Diamond Light Source® and the Diamond logo are registered trademarks of Diamond Light Source Ltd
Registered in England and Wales at Diamond House, Harwell Science and Innovation Campus, Didcot, Oxfordshire, OX11 0DE, United Kingdom. Company number: 4375679. VAT number: 287 461 957. Economic Operators Registration and Identification (EORI) number: GB287461957003.