___________________________________
Industrial Liaison Group:
Tel: +44 (0) 1235 778797
E-mail: [email protected]

Crystallography is the experimental science of determining the arrangement of atoms in a crystalline solid, which can be found everywhere in nature; from salt to snowflakes to gemstones.
X-ray crystallography is currently the most favoured technique for structure determination of proteins and biological macromolecules and for determining the structure of a molecular material. Increasingly, researchers from all branches of science require structural information to shed light on previously unanswered questions. Furthermore, in the case of biological sciences, the availability of a protein structure can provide a more detailed focus for future research and the development of drug targets.
Crystallographers use the properties and inner structures of crystals to determine the arrangement of atoms and generate knowledge that is used by chemists, physicists, biologists, and others. Within the past century, crystallography has been a primary force in driving major advances in the detailed understanding of materials, synthetic chemistry, the understanding of basic principles of biological processes, genetics, and has contributed to major advances in the development of drugs for numerous diseases.
In 1912, Max von Laue and colleagues at the University of Munich, Germany came up with the idea to send a beam of X-rays through a copper sulphate crystal and record the results on photographic plates. He persuaded his colleagues Walter Friedrich and Paul Knipping, both of whom had more practical experience with X-rays than von Laue himself, to perform the experiment, the results of which showed diffraction spots surrounding the central spot of the primary beam.
The discovery came 17 years after Wilhelm Conrad Röntgen had first demonstrated the existence of X-rays and their nature was still undetermined. Physicists suspected that X-rays were a form of electromagnetic radiation, but had been unable to obtain solid evidence for their diffraction. Von Laue's experiment presented evidence for both the wave nature of X-rays and the space lattice of crystals at the same time as the diffraction spots were caused by X-rays impinging on a regular array of scatterers, in this case, the repeating arrangement of atoms within the crystal. The scatterers produced a regular array of spherical waves which gave the bright spots on the photographic plate.
Within a year of this discovery, in 1912, William Henry Bragg and son Lawrence had exploited the phenomenon to solve the first crystal structure and create a mathematical formula, Bragg’s Law, which showed how to work out the atomic structure of a sample based on the diffraction pattern it produced when exposed to X-rays. The discoveries of von Laue and Bragg gave birth to two new sciences, X-ray crystallography and X-ray spectroscopy, and resulted in two Nobel Prizes: Max von Laue “for his discovery of the diffraction of X-rays by crystals” in 1914 and to Sir William Lawrence Bragg and his father, Sir William Henry Bragg, “for their services in the analysis of crystal structure by means of X-rays” in 1915. Crystallography has to date, directly or indirectly, produced the largest number of Nobel Laureates throughout history, up to now 29 Prizes for 48 Laureates..
The Braggs also mentored a dream team of crystallographers who went on to work out the structures of a huge range of molecules. From Kathleen Lonsdale, JD Bernal, Dorothy Hodgkin, to David Phillips, John Kendrew, and Max Perutz. Also, Rosalind Franklin and others even helped map the structure of DNA, probably the most famous result of X-ray crystallography.
 The Braggs carried out their early experiments with the first spectrometer: a small machine capable of creating a diffraction pattern from X-rays. H
The Braggs carried out their early experiments with the first spectrometer: a small machine capable of creating a diffraction pattern from X-rays. H
However, this process was laborious and fraught with difficulty. These days, scientists have access to synchrotrons like Diamond, capable of reliably producing light 10 billion times brighter than the sun. This high-quality light is something the Braggs could only have dreamed of, and it means that what once took scientists years, can now be done in minutes.
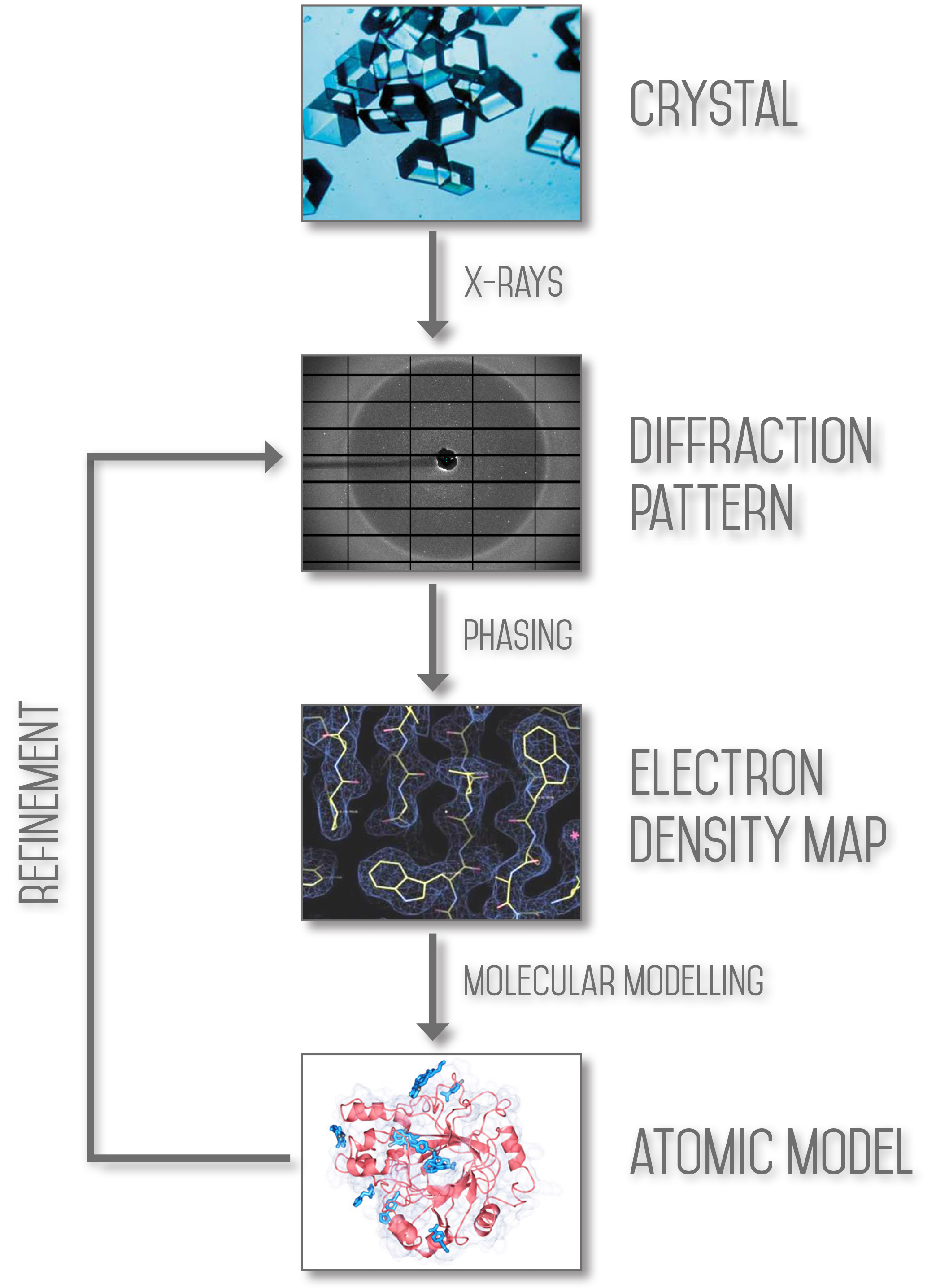
The aim of X-ray crystallography is to obtain a three dimensional molecular structure from a crystal. A purified sample at high concentration is crystallised and the crystals are exposed to an X-ray beam, usually under cryogenic conditions to protect the sample from radiation damage. The resulting diffraction patterns can then be processed, initially to yield information about the crystal packing symmetry and the size of the repeating unit that forms the crystal. This is obtained from the pattern of the diffraction spots.
The intensities of the spots can be used to determine the “structure factors” from which a map of the electron density can be calculated. Various methods can be used to improve the quality of this map until it is of sufficient clarity to permit the building of the molecular structure (using the protein sequence in the case of macromolecular crystallography). The resulting structure is then refined to fit the map more accurately and to adopt a thermodynamically favoured conformation.

The growth of protein crystals of sufficient quality for structure determination is, without doubt, the rate limiting step in most crystallographic work, and is the least well understood. The principle of crystallisation, whether of macromolecules or salts is to take a solution of the sample at high concentration and induce it to come out of solution; if this happens too fast then precipitation will occur, but under the correct conditions crystals will grow.
The elucidation of these conditions determines the rate limiting step and indeed whether or not the project will be possible. Many projects prove not to be possible because of the inability to crystallise the material or protein. The magnitude of the problem can be understood when one considers all of the variables involved: the choice of precipitant, its concentration, the buffer, its pH, the protein concentration, the temperature, the crystallisation technique, and the possible inclusion of additives.
X-ray Crystallography can be divided into two main areas at Diamond; Macromolecular Crystallography (MX), a technique used to study biological molecules such as proteins, viruses and nucleic acids (RNA and DNA), and Small Molecule X-ray Crystallography, a technique key to determining the structure of small and weakly diffracting molecules.

Macromolecular Crystallography (MX, also referred to as Protein Crystallography or PX) is the most powerful method for determining the atomic three dimensional structures of large biological molecules. It is a vital tool for linking structure with function, for rational drug design, for investigating protein folding and for relating other structural information, such as evolutionary relationships, from biological molecules.
The MX beamlines here at Diamond allow structure determination by molecular replacement, where the structure of a related molecule is already known, by standard isomorphism replacement with heavy atoms and by Single wavelength Anomalous Dispersion (SAD) or Multiwavelength Anomalous Dispersion (MAD) measurements, in which the wavelength of the X-rays is tuned to be near or at the natural absorption wavelength of selected atoms in the macromolecule. This enables the detection of small differences in the diffraction pattern for reflections that are related as Friedel pairs and these differences allow phase determination to determine the structure through crystallographic methods.
Macromolecules tend to form small, imperfect and weakly diffracting crystals. The high brightness of the X-rays generated at Diamond makes the collection of precise measurements possible. In addition, the state of the art MX beamlines are fitted with advanced robotic systems and software for automated sample handling, crystal centring and data collection along with rapid data processing.
Applications of MX include rational drug design, enzyme mechanisms, supramolecular structure, molecular recognition, nucleic acids, structural genomics and high throughput crystallography. Crystallography experiments at Diamond have recently yielded a potential vaccine for foot-and-mouth disease and have helped scientists to study T-cells: a natural defence mechanism in the body which could provide an effective treatment against cancer.
Single-crystal X-ray diffraction techniques are considered the definitive means of determining the structure of a molecular material. It is an indispensable pillar of synthetic chemistry as it provides conclusive confirmation that a target material has been formed. X-ray crystallography, using single-crystal methods, also provides the most accurate means of following the response of a molecular structure to an external influence such as variation in pressure and temperature, photo-excitation or, for porous systems, the exchange of a gas.
The high photon flux offered by the I19 beamline, a dedicated instrument for small molecule crystallography, here at Diamond allows crystal structure studies of samples that are too weakly scattering, due to either their size or the complexity of their underlying molecular structure, to be successfully carried out using any other X-ray source, for example laboratory-based diffractometers.
Small molecule X-ray crystallography techniques underpin chemistry and materials science in strategically important areas, such as pharmaceuticals to assess the stability of potential drug candidates, catalysis to observe real time changes to variable environmental conditions, and in gas storage to research metal-organic frameworks for hydrogen and fuel gas storage.
Would you like to know more about crystallography and how you can apply it to your research? Do you perhaps have a structural problem that you are unable to solve in your lab or a material you wish to find out more about? Then please get in touch with the Industrial Liaison Team at Diamond.
The Industrial Liaison team at Diamond is a group of professional, experienced scientists with a diverse range of expertise, dedicated to helping scientists and researchers from industry access the facilities at Diamond. We’re all specialists in different techniques and have a diverse range of backgrounds so we’re able to provide a multi-disciplinary approach to solving your research problems. We offer services ranging from full service; a bespoke experimental design, data collection, data analysis and reporting service through to providing facilities for you to conduct your own experiments.
We’re always happy to discuss any enquiries or talk about ways in which access to Diamond’s facilities may be beneficial to your business so please do give us a call on 01235 778797 or send us an e-mail. You can keep in touch with the latest development by following us on Twitter @DiamondILO or LinkedIn.
Diamond Light Source is the UK's national synchrotron science facility, located at the Harwell Science and Innovation Campus in Oxfordshire.
Copyright © 2022 Diamond Light Source
Diamond Light Source Ltd
Diamond House
Harwell Science & Innovation Campus
Didcot
Oxfordshire
OX11 0DE
Diamond Light Source® and the Diamond logo are registered trademarks of Diamond Light Source Ltd
Registered in England and Wales at Diamond House, Harwell Science and Innovation Campus, Didcot, Oxfordshire, OX11 0DE, United Kingdom. Company number: 4375679. VAT number: 287 461 957. Economic Operators Registration and Identification (EORI) number: GB287461957003.