 | ||
Controlling defects in metal-organic frameworks |
Metal-organic frameworks (MOFs) are versatile materials with many applications in areas such as catalysis and hydrogen storage. Recently it was found that certain MOFs contain defects, which could lead to new functionality if the locations of the defects are better understood so that they can be exploited. Correlated disorder and defects are abundant in exotic materials such as ferroelectrics and superconductors, where the defects are not randomly distributed but interact such that their positions are dependent on other defects. Previous research has indicated that one of the canonical MOFs, UiO-66(Hf), contains ligand-absence defects. The Extreme Conditions beamline (I15) at Diamond Light Source has the capacity to measure anomalous scattering from hafnium (Hf), and so was able to confirm that UiO-66(Hf) can contain not only ligand-absence defects, but also correlated Hf cluster absences.
Beamline I15 Scientific Highlight
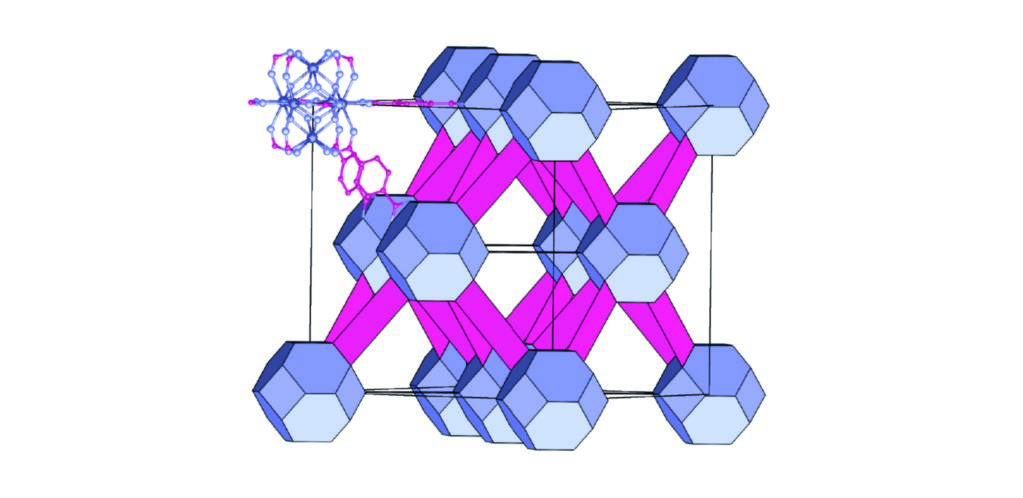
Figure 1: The structure of non-defective fcu UiO-66(Hf). A single Hf6(OH)4O4(O2C)12 cluster is shown in full in the top left of unit cell, with the rest of the unit cell represented as polyhedra. The Hf clusters are represented by blue truncated octahedra and the bdc2- ligands by pink rectangles. The atomic colour scheme is as follows: Hf, dark blue; O, light blue and C, pink. H atoms are omitted for clarity.
Point defects, and the local fields they cause, are crucial for many materials’ properties, as they allow for the creation of correlated states with unusual properties, for example relaxor ferroelectrics1, colossal magnetoresistance2, and the correlated motion of ions through superionic conductors3. Despite the importance of defects in conventional materials, it is only recently that the role that defects could play in the chemistry of MOFs has begun to be appreciated. Recent work has now established that it is possible to dope MOFs with large numbers of ligand-absence defects. These defective MOFs have been shown to have significantly enhanced both guest capacities4 and catalytic activity5. For some MOFs, including the canonical MOF UiO-66, these ligand-absences are present even under standard reaction conditions. UiO-66 is assembled from of MIV 6(OH)4O4 clusters (M = Zr,Hf) linked by benzene-1,4-dicarboxylate (H2bdc) linkers into a face centred cubic structure (Fig. 1), and its combination of high connectivity and strong metal-ligand bonding means that it is one of the most stable MOFs. The concentration of defects in UiO-66 can be regulated by altering the concentration of ‘modulators’ – monotopic acids, typically acetic, benzoic or trifluoroacetic acids – added to the reaction mixture. In all these reported cases of ligand-absence defects, the presence of correlations was not observed.

Figure 2: (a) The low angle region powder X-ray diffraction patterns of UiO-66(Hf), for samples containing different defect orderings, from top to bottom, nano-reo UiO-66(Hf), experimental and calculated from a supercell model, experimental and calculated nondefective fcu UiO-66(Hf) and defective ordered reo UiO-66(Hf); (b) Measurement of experimental powder diffraction patterns at the Hf K-edge (65.3 keV) and away from it (55 keV) revealed a subtle change in the relative intensities of the broad superlattice reflections, indicating that scattering from Hf is contributing to the diffuse scattering.
Source publications:
Cliffe, M. J., Wan, W., Zou, X., Chater, P. A., Kleppe, A. K., Tucker, M. G., Wilhelm, H., Funnell, N. P., Coudert, F. –X. & Goodwin, A. L. Correlated defect nanoregions in a metal-organic framework. Nature Communications 5, doi: 10.1038/ncomms5176 (2014).
References:
1. Xu, G. Y., Zhong, Z., Bing, Y., Ye, Z. G. & Shirane, G. Electric-field-induced redistribution of polar nano-regions in a relaxor ferroelectric. Nature Materials 5, 134-140, doi:10.1038/nmat1560 (2006).
2. Adams, C. P., Lynn, J. W., Mukovskii, Y. M., Arsenov, A. A. & Shulyatev, D. A. Charge ordering and polaron formation in the magnetoresistive oxide La0.7Ca0.3MnO3. Physical Review Letters 85, 3954-3957, doi:10.1103/ PhysRevLett.85.3954 (2000).
3. Miller, T. A. et al. The mechanism of ultrafast structural switching in superionic copper (I) sulphide nanocrystals. Nature Communications 4, doi:10.1038/ncomms2385 (2013).
4. Park, J., Wang, Z. U., Sun, L.-B., Chen, Y.-P. & Zhou, H.-C. Introduction of Functionalized Mesopores to Metal-Organic Frameworks via Metal- Ligand-Fragment Coassembly. Journal of the American Chemical Society 134, 20110-20116, doi:10.1021/ja3085884 (2012).
5. Vermoortele, F. et al. Synthesis Modulation as a Tool To Increase the Catalytic Activity of Metal-Organic Frameworks: The Unique Case of UiO-66(Zr). Journal of the American Chemical Society 135, 11465-11468, doi:10.1021/ja405078u (2013).
Funding acknowledgements:
M.J.C. and A.L.G. acknowledge financial support from the E.P.S.R.C. (EP/G004528/2) and the E.R.C. (Grant Ref: 279705. X.Z. and W.W. acknowledge financial support from the Swedish Research Council (VR), the Swedish Governmental Agency for Innovation Systems (VINNOVA) and the Knut and Alice Wallenberg (KAW) Foundation through the project grant 3DEM-NATUR. The EM facility is supported by the KAW Foundation. This work was performed using HPC resources from GENCI-IDRIS (grant i2014087069).
Corresponding author:
Matthew Cliffe, University of Oxford, [email protected]


 A brighter light for science
A brighter light for science
