The challenge
The hunt for viable green alternatives to traditional petrol and diesel engines has led a move towards natural gas engines, which produce less carbon dioxide emissions.
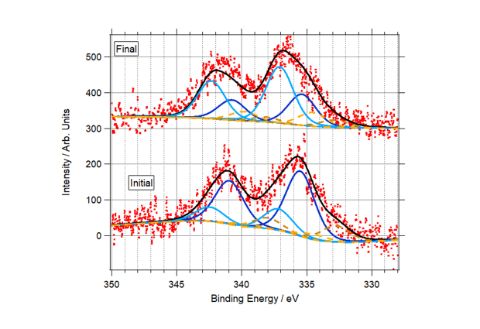
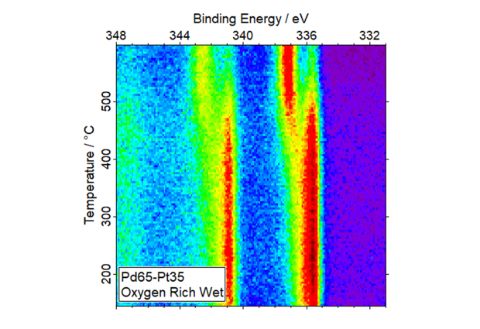
But natural gas engines pose other problems due to unburnt methane (a potent greenhouse gas) in the exhaust feed. So scientists at Diamond, Johnson Matthey – a global leader in sustainable technologies - and the University of Reading, have been researching ways to improve catalysts to convert residual methane into more environmentally friendly products.