____________________________________
Industrial Liaison Group:
Tel: +44 (0) 1235 778797
E-mail: industry@diamond.ac.uk
T-cell receptors (TCRs) are protein complexes found on the surface
of human T-cells responsible for recognising a large variety of
antigen peptides. When a TCR binds to a specific antigen it triggers
an immune response that is part of the adaptive immune system
and prevents the growth of pathogens.
There is great interest in using this natural defence system for
cancer immunotherapy by producing modified TCRs that bind
cancer antigens and activate an immune response against
tumours.
In order to understand what characteristics of peptide antigens are important for TCR response and selectivity, scientists from Immunocore and GlaxoSmithKline focused on three newly identified human TCRs that bind to a known cancer antigen peptide.
Although all three TCRs bind to the same cancer antigen, their specificity profiles are different. To gain insight into how TCRs with different specificities can bind to the same antigen, the researchers needed high-resolution structural data showing the TCR-peptide binding interface.
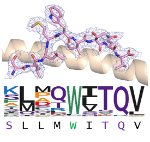
The TCRs of interest were co-crystallised with the cancer antigen peptide, and X-ray diffraction data were collected at Diamond’s state of the art MX beamlines. The data was used to determine the atomic resolution structures of the TCRs bound to antigen peptides.
The structural analysis shows that although the TCRs bind to the same peptide sequence, they recognise different conformations of the peptide. This level of structural detail explains how TCRs can use different binding modes but still be specific for the same peptide antigen.
The highly intense synchrotron X-ray radiation and the advanced data collection capabilities of Diamond’s MX beamlines were critical to determine the structures of the TCR-peptide complexes at atomic resolution that enabled the scientists to observe the different TCR binding modes. Understanding how these receptors use different structural features to acquire specificity for the same antigen is important for the development of future cancer immunotherapies.

.jpg)
.png)
“TCRs naturally bind their pHLA ligand weakly and require significant engineering to increase affinity whilst maintaining specificity. Determining co-complex crystal structures helps triage molecules and aids the engineering process. The MX beamlines, robotics and software at Diamond enable us to rapidly screen crystals and collect the data we require.”
Dr Ross Robinson, Group Leader, Immunocore


Diamond Light Source is the UK's national synchrotron science facility, located at the Harwell Science and Innovation Campus in Oxfordshire.
Copyright © 2022 Diamond Light Source
Diamond Light Source Ltd
Diamond House
Harwell Science & Innovation Campus
Didcot
Oxfordshire
OX11 0DE
Diamond Light Source® and the Diamond logo are registered trademarks of Diamond Light Source Ltd
Registered in England and Wales at Diamond House, Harwell Science and Innovation Campus, Didcot, Oxfordshire, OX11 0DE, United Kingdom. Company number: 4375679. VAT number: 287 461 957. Economic Operators Registration and Identification (EORI) number: GB287461957003.