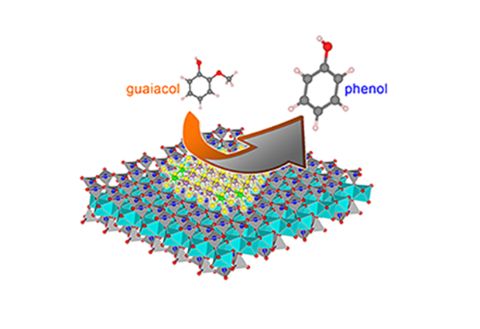
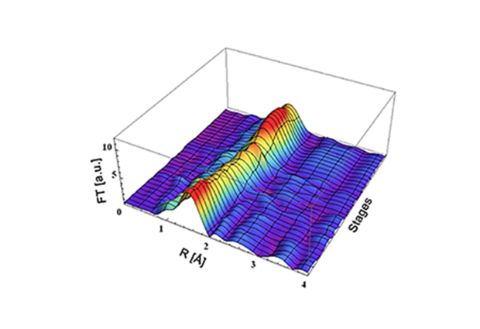
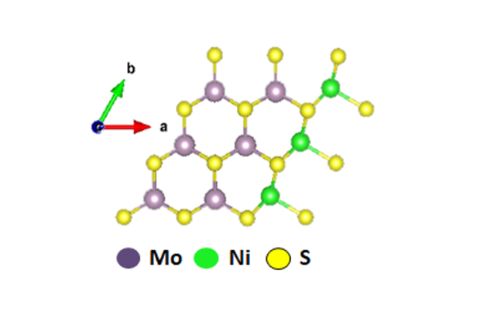
Designing an effective catalyst to convert palm oil waste into sustainable biofuels
Indonesia is one of the largest suppliers of palm oil in the world, producing 42 million tonnes in 20181. It is also experiencing an increase in car usage, coupled with a growth in imports of fuel.
To overcome this problem, the Indonesian government is driving a move to biofuels. Until recently the fresh fruit bunch from palm oil has successfully been used, however the empty fruit bunch (EFB) and palm kernel shell (PKS) provide a more sustainable source of lignocellulose, a key component in second generation biofuel production.
One prospective method for the biofuel production is conversion of lignocellulose into bio-oil via fast pyrolysis and then upgrading the bio-oil over a catalyst, to remove oxygen. However, the existing alumina-based and noble metal catalysts still suffer from catalyst deactivation due to carbon deposition and metal leaching.