The Solution
The study uses Fe‑based catalysts dispersed onto two chemically different supports: O and N‑doped graphite, to determine the influence of the carbon surface chemistry on the structural and catalytic properties of these materials in the CO2 reduction reaction. The catalysts were tested under different potential conditions in a low concentration bicarbonate solution, by means of electrochemical techniques, to identify conditions for maximal efficiency as well as changes in selectivity.
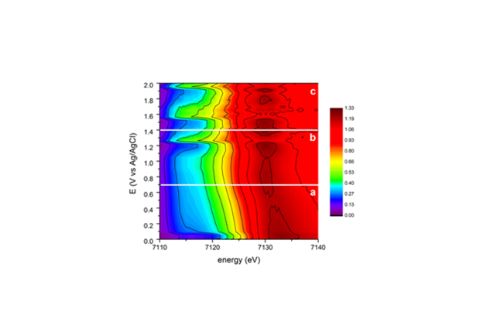
By using Fe K‑edge X‑ray absorption spectroscopy and gas‑chromatography, the scientists were able to study the catalysts in situ during CO2 reduction and Hydrogen Evolution Reaction (HER), and thus to correlate the structural changes and the products formed.
The results of the study showed that N‑dopants in graphite stabilise Fe(II) species, thus hindering their further reduction to metallic Fe at potentials relevant to acetic acid production. In contrast, Fe supported on O‑doped graphite was less favourable for CO2 production, but did produce H2 in the entire range of potentials investigated. A correlation between the production of H2 from water electrolysis and the reduction of initial Fe(III) to a metallic state was observed. Despite its poor performance in the CO2 reduction reaction, this catalyst presented itself as a good candidate for substituting precious metals for HER.