Welcome to I04-1
I04-1 is a fixed wavelength monochromatic beamline, with its undulator in I04's straight section. The beamline is aimed at high-throughput data collection for well-diffracting crystals, with off-the-shelf robotics and a stable beam thanks to a simple beamline design. Flux has been significantly increased since the insertion device install in July 2022 offering 3.8x1012 (photons/second) with the 70um apeture.
As of April 2015, the beamline is also offering a world-first, namely routine medium-throughtput fragment screening by crystal structure, including all steps from crystal soaking and harvesting to data analysis. Full details and application process are here.
WEBCAMS
- Live view! (intranet)Goniometer and Experiment Overview
Background
I04-1 delivers a high-throughput approach similair to that pioneered in industry, where it is axiomatic that the best way to study ligand binding involves developing a well-diffracting crystal system, which allows for the rapid X-ray data collection necessary to observe the binding of the many compounds generated by medicinal chemistry. The implication for a beamline is that flexibility is less important than stability and speed. I04-1 is heavily utilised by the XChem platform, as well as being fully availiable to the MX user community for rotational cryogenic experiments.
Research program
The research focus is on taking high-throughput approach to its logical extreme, namely all aspects of implementing and exploring routine medium- throughtput fragment screening by crystal structure. This involves a lot more than just the X-ray experiment, and since late 2012 the beamline has been partnered with the Protein Crystallography group, now at the Centre for Medicince Discovery in University of Oxford.
Developments include:
- Academic and Industrial access to high throughput crystallogrpahic screening
- Rapid synthesis of compounds using automated robotics platforms
- Fragalysis platform - Quick review of Crystallographic fragment data for advancing medicinal chemistry of screened targets
- Development of algorithms for hit identifentifaction in crystallographic screens: Pan-Dataset Density Analysis (PanDDA)
- Development of room temperature crystallographic fragment screening
I04-1 Specification
| Energy | fixed, monochromatic: 0.920Å / 13.53 keV |
| Experimental phasing | SAD: optimal for Br, good for Se |
| Flux (ph/sec) |
3.8 x 1012 (70um aperture, 300mA ring current) 2.5 x 1012 (50um aperture, 300mA ring current) 1.3 x 1012 (30um aperture, 300mA ring current) 9.0 x 1011 (20um aperture, 300mA ring current) 3.0 x 1011 (10um aperture, 300mA ring current) |
|
Beam size options (µm): |
|
| Detector | Eiger 2 XE 9M |
| Maximum resolution (Å) | 1.6 |
| Dataset time, typical | 7.2s (3600 Images, 0.1 degrees, 100% Transmission, 0.002s) |
| Sample changer | Diamond BART with unipucks |
| Sample exchange time | < 20s |
| No. of pins in dewar | 592 |
| Pins and Pucks | SPINE standard pins (only!) and Unipucks |
|
Samples rate (xtals/hour) |
15-30 (manual, user centring) 29 (queued, automatic X-ray centring) |
Please discuss your requirements with a member of the beamline team before your experiment.
SAD phasing at I04-1
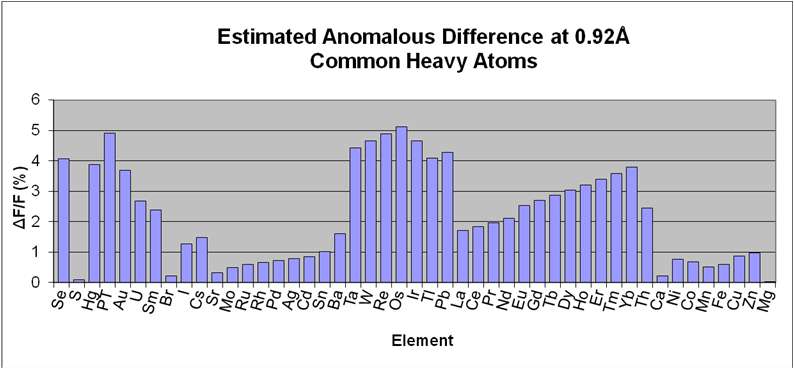
I04-1's wavelength is fixed at the Bromine K edge (E = 13530 eV, λ = 0.9163 Å), where a strong anomalous signal can be measured for most commonly-used heavy atom derivatives (see figure). SAD is usually very effective here - unsurprisingly: thanks to modern detectors and phasing algorithms, tunability is essential only for special cases, especially if the experiment is done carefully (see e.g. Krojer, Pike, von Delft, Acta D 2013)
Estimated anomalous difference at 0.92Å resolution for common elements. These were calculated assuming: for all elements, 1 fully occupied heavy atom site per 300 residues; for Se, 1:42 heavy atoms:residue; and using the formula derived from Smith, J., Curr. Opin. Struct. Biol. vol1, p1002 (1991).


 Industrial Liaison Office
Industrial Liaison Office
.png)