________________________________________
Industrial Liaison Group:
Tel: +44 (0) 1235 778797
E-mail: industry@diamond.ac.uk
____________________________________
Diamond is a world leading facility for structural biology. The outstanding design of the macromolecular crystallography (MX) beamlines combines the provision of cutting edge technical instruments essential for de novo structure solution and the study of protein ligand interactions with the latest developments in automation required to accelerate lead identification and ligand screening processes.
Our state of the art MX beamlines are fitted with advanced robotic systems and software for automated sample handling, crystal centring and data collection. In addition, microfocus beamlines are perfectly suited for studying small or disordered crystals and facilities for containment level 3 samples are in place.
At Diamond, we have developed high performance automated processing pipelines for integration, scaling and phasing data in real time for immediate structure analysis directly at the beamline.
We follow a programme of continuous upgrades to maintain our world leading position for structural biology research. Beamline specifications are updated frequently as upgrades are introduced onto the beamlines, so check for our very latest specifications in the table below.
Complementary techniques for structural biology are also available at Diamond including protein small angle scattering (SAXS), circular dichroism and a fragment screening platform based on X-ray crystallography.
| Beamline | I03 | I04 | I04-1 | I24 |
| Flux (ph/s) at standard energy | 1.7 x 1012 | 2.5 x 1011 | 9.0 x 1011 | 3.0 x 1012 |
| Focused beam size (HxV, µm) | 90 x 20* | 5 x 10 - 110 x 100 | 60 x 50 | 5 x 5 - 50 x 40 |
| Detector type/Frequency (Hz) | Pilatus3 6M 100 | Pilatus 6M 25 | Pilatus 6M 25 | Pilatus3 6M 100 |
| Sample changer | BART | BART | BART | BART |
| Sample exchange time (s) | 18 | 18 | <20 | <20 |
| Dewar capacity (pins) | 480 | 592 | 592 | 592 |
| Throughput (samples/hour) | 15-25 | 15-25 | 15-35 | 15-25 |
| Energy (keV) | 21 -5.2 | 18.0 - 6.0 | 13.5 | 20.0 - 6.4 |
| Standard energy (keV) / wavelength (Å) | 12.7 / 0.98 | 12.7 / 0.98 | 13.5 / 0.92 | 12.8 / 0.97 |
| Microfocus beam | No | Yes | No | Yes |
| Mini beam aperture | Yes | No | Yes | No |
| Remote crystal annealing | Yes | Yes | Yes | Yes |
| Plate screening | Yes | No | No | Yes |
* Defocused and microfocused beam options are also available.
High throughput screening for ligand optimisation, cryo-optimisation and RT data collection in SBS-format crystallisation plates.
Dehydrate / rehydrate samples whle monitoring their diffraction properties under humidity control.

Grid scan provides a precise tool for location and/or centring of small or inhomogeneous crystals. Helical scan maximises completeness and quality of data from small elongated crystals and needles.

The speed and spatial resolution of our detectors allow used to resolve diffraction spots from crystals of very large unit cells such as multi-protein complexes and membrane proteins.
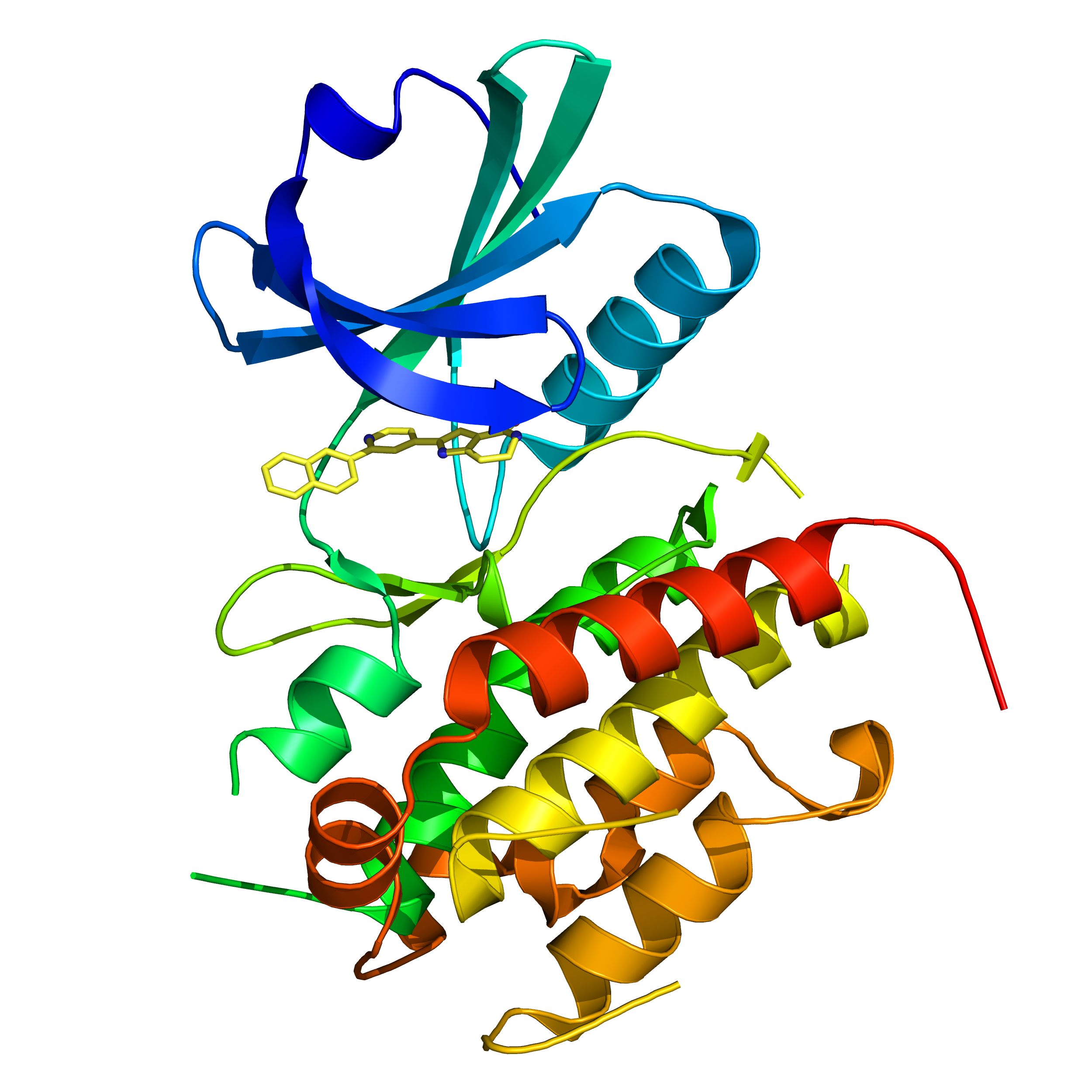
Programmed Cell Death Protein 1 (PD1) is a receptor protein on the surface of T cells which is involved in the suppression of the immune system. Blocking PD1 has been a successful approach to enhance anti-cancer immune responses, and there is also significant interest in developing molecules that can trigger PD1 signalling to treat autoimmune diseases.
Read more...
A group of enzymes called β-Glucosidases are used in the food industry to hydrolyse glycosidic bonds in complex sugars. Since many food industrial processes involve harsh conditions like high concentrations of solvents and sugars, low pH, and high temperatures, there is a need for these kinds of enzymes to be adapted to function in extreme environmental conditions.
These enzymes can be employed for the cleavage of isoflavone glucosides from soy flour, to produce soy isoflavones, a valuable food supplement.

T-cell receptors (TCRs) are protein complexes found on the surface
of human T-cells responsible for recognising a large variety of
antigen peptides. When a TCR binds to a specific antigen it triggers
an immune response that is part of the adaptive immune system
and prevents the growth of pathogens.
There is great interest in using this natural defence system for
cancer immunotherapy by producing modified TCRs that bind
cancer antigens and activate an immune response against
tumours.

Membrane proteins represent a large group of important drug targets with over 50% of approved drugs targeting these proteins within the body.
Understanding the 3D structure of membrane proteins is invaluable for target discovery, validation, and for hit optimisation. X-ray macromolecular crystallography (MX) is a principal research method for acquiring structural information on membrane proteins, but for this to be successful we require the production of good quality crystals from the target protein, or its complexes with potential drug candidates.
It is particularly difficult to produce and purify membrane proteins in sufficient yield and quality for crystallisation, and furthermore, membrane protein crystallisation itself represents a significant challenge.
Respiratory infections are one of the biggest killers in the western world, and pneumococcal infections can range from mild to potentially deadly. Children and the elderly are most at risk, as well as those with compromised immune systems. There are approximately 90 distinct pneumococcal serotypes and, although not all cause disease, this makes treatment challenging. An effective conjugate vaccine has been available since 2001, however there has been a rise in antibiotic resistance in serotypes not covered by this conjugate vaccine and previously non-infectious serotypes are increasing in virulence. This makes treatment of pneumococcal pneumonia and less severe illnesses such as otitis media more difficult to manage. Therefore finding new approaches to combat pneumococcal disease remains a high priority.
Read more...
Influenza viruses altogether affect 5%-10% of the adult population and 20-30% of children each year. These annual epidemics are estimated to result in about approximately 3 to 5 million cases of severe illness, leading to between 250,000 to 500,000 deaths around the world. Integral to viral replication, polymerases are enzymes that copy the viral genome and produce messenger RNA which is then used to make building blocks for new virus particles.
Read more...
As fast Pilatus detectors have become the gold standard at Diamond’s MX beamlines, the time needed for collecting a complete dataset is now less than two minutes. This gives a user very limited time to process data and subsequently decide what would be the best strategy for collection of data from remaining crystals.
Read more...
In the past two decades, fragment based drug discovery has emerged as a powerful method to discover and develop new drugs. Such strategy relies on the identification of low molecular weight compounds that bind weakly to a target. The structural information gained on fragment-target complexes provides an excellent starting point for the development of potent and selective drugs.
Read more...
Methylxanthines, including caffeine, theophylline (found in tea), and theobromine (a constituent of chocolate) are among the most widely consumed substances in the world, but their effects on the brain are not fully understood. Previously published research has shown a correlation between higher intake of caffeine and lower incidence of Parkinson’s disease. Adenosine A2A receptors regulate the effects of neurotransmitters in the brain, cardiovascular and immune systems, and are of particular interest as a drug target.
Read more...
Fragment-based and structure-based drug discovery programmes put a strong emphasis on frequent routine macromolecular crystallography data collection from numerous samples. Other types of projects may only need occasional data collection on a few samples or researchers may need to run an experiment they have not done before.
Read more...
X-ray structural investigation of macromolecules (proteins, viruses, enzymes etc) is a complex and time consuming process where the quality of information obtained about the three-dimensional structure is strongly dependent on the quality of crystals produced. Therefore, the structures of many important targets of pharmaceutical and biotechnology industry remain unknown simply because researchers are unable to obtain crystals of sufficiently high quality or size.
Read more...
Huntington’s disease (HD) is a dominantly-inherited neuropsychiatric disorder which causes severe motor dysfunction, depression and dementia. It has been known since 1993 that the disease is due to mutation of a single gene coding for huntingtin (HTT) that extends the poly-glutamine (poly-Q) repeats in the protein. Inhibiting the enzyme responsible for the cleavage of mutant HTT - thereby reducing the generation of the poly-Q fragments - could be one way of preventing neurodegeneration. Caspase-6 was identified as this processing enzyme, yet, while structures of other caspase enzymes have been studied, caspase-6 has been suggested to be this processing enzyme but, while structures of other caspase enzymes have been solved.
Read more...
Macromolecular crystallography (MX) is a key asset in the development of new drugs via structure based drug discovery. This process is now widely used by the pharmaceutical industry to gain structural insights into drug targets and map interactions with ligands. However, isolating and optimising a new lead compound is a time consuming process which usually involves screening hundreds of samples.
Read more...
Developments on macromolecular crystallography (MX) beamlines have achieved very high throughput and microfocus X-ray facilities have made it possible to successfully collect diffraction data from samples previously considered too small or too disordered. However, when the use of microfocus beams is coupled with the small size and poor optical properties of some samples, sample alignment and detection becomes a key problem.
Read more...
The size and diffraction quality of crystals remain the major limiting factors of macromolecular crystallography (MX). Many crystals of challenging targets are small, weakly diffracting and indeed extremely sensitive to manual handling. The time consuming and often damaging process of cryo-protecting, mounting and cryocooling of crystals can significantly decrease the crystal order, resolution and overall quality of the data.
Read more...
Investigating the 3D structure of macromolecules by X-ray crystallography requires the production of high quality crystals, and yet this step remains a tedious and empirical task. Weak packing and high solvent content are among the most frequent factors causing a negative impact on the acquisition of suitable diffraction images. It is, therefore, useful to develop post-crystallisation methods to enhance crystal quality.
Read more...
Fragment-based and structure-based drug discovery projects require regular and frequent proprietary access to macromolecular crystallography (MX) beamlines at synchrotron facilities. However, geographic location, shutdown periods, limited human resources and beamtime availability can restrict the access to such facilities.
Read more...Diamond Light Source is the UK's national synchrotron science facility, located at the Harwell Science and Innovation Campus in Oxfordshire.
Copyright © 2022 Diamond Light Source
Diamond Light Source Ltd
Diamond House
Harwell Science & Innovation Campus
Didcot
Oxfordshire
OX11 0DE
Diamond Light Source® and the Diamond logo are registered trademarks of Diamond Light Source Ltd
Registered in England and Wales at Diamond House, Harwell Science and Innovation Campus, Didcot, Oxfordshire, OX11 0DE, United Kingdom. Company number: 4375679. VAT number: 287 461 957. Economic Operators Registration and Identification (EORI) number: GB287461957003.