___________________________________
Industrial Liaison Group:
Tel: +44 (0) 1235 778797
E-mail: industry@diamond.ac.uk
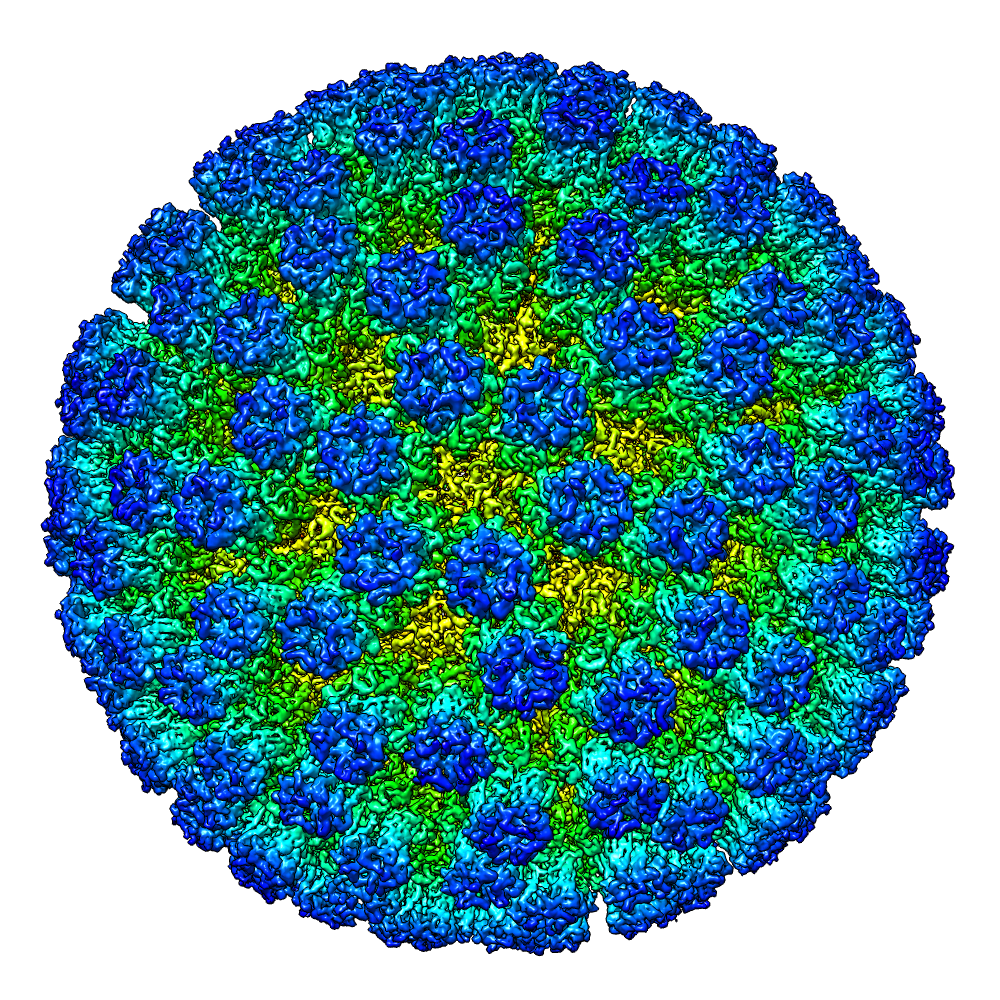
The Electron Bio-Imaging Centre (eBIC) was established at Diamond following the award of a £15.6 million grant from the Wellcome Trust, the Medical Research Council (MRC) and the Biotechnology and Biological Sciences Research Council (BBSRC). The location of eBIC enables scientists to combine their techniques with many of the other cutting-edge approaches that Diamond offers. eBIC provides scientists with state-of-the-art experimental equipment and expertise in the field of cryo-electron microscopy, for both single particle analysis and cryo-tomography. Currently eBIC houses four Titan Krios microscopes, a Talos Arctica and a Scios cryo-FIB/SEM. The Polara microscope is located at the University of Oxford.
Single particle cryo-electron microscopy
This technique requires the collection of a large number of movies from mono-disperse protein complexes or viruses such that their 3D structure can be determined. The Titan Krios and Polara microscopes are equipped with direct electron detectors and automated data collection software that allow a large number of movies to be collected from single particle samples. The type of single particles that can be imaged range from small protein complexes (150 kDa) to large viruses (2 MDa). For proteins smaller than 150 kDa the use of the phase plate may be required and should be discussed with eBIC staff beforehand.
Molecular cryo-electron tomography
This technique can be used with single particle samples and is excellent at generating initial models for single particle analysis or for analysing repeating structures in larger pleomorphic structures. Tilt series are collected at areas of interest, which can then be aligned and reconstructed to generate 3D reconstructions. For thicker specimens zero-loss imaging is recommended.
Cellular cryo-electron tomography
This technique is used to look at large pleomorphic objects such as vesicles, isolated organelles, bacteria, and intact eukaryotic cells. Tilt series are collected at areas of interest which can then be aligned and reconstructed to generate a 3D reconstructions. For thicker specimens zero-loss imaging is recommended up to a maximum sample thickness is of ~0.5 um.
Electron crystallography (coming soon)
This technique requires thin 3D crystals or 2D crystals of lipid-embedded membrane proteins. For thin 3D crystals, electron diffraction data are collected. For 2D crystals, both projection images and electron diffraction data (if feasible) are collected and then combined to generate a 3D reconstruction of the protein of interest.
 Titan Krios I, II, III and IV are state of the art 300 KeV cryo-electron microcopes from FEI. They are equiped with the latest generation of direct electron detectors, the FEI Falcon II and the Gatan Bio-quantum K2 summit. Both detectors are intergated into FEI's automated data aquisition softwares for single particle and tomography, EPU and TOMO4 respectively.
Titan Krios I, II, III and IV are state of the art 300 KeV cryo-electron microcopes from FEI. They are equiped with the latest generation of direct electron detectors, the FEI Falcon II and the Gatan Bio-quantum K2 summit. Both detectors are intergated into FEI's automated data aquisition softwares for single particle and tomography, EPU and TOMO4 respectively.
 The Talos Arctica is FEI’s new generation fully automated 200 kV FEG electron microscope. It is equipped with an autoloader system that permits loading of up to twelve specimens, automated data collection at liquid nitrogen temperature, and features a Constant-PowerTM objective lens for optimal contrast/resolution balance.
The Talos Arctica is FEI’s new generation fully automated 200 kV FEG electron microscope. It is equipped with an autoloader system that permits loading of up to twelve specimens, automated data collection at liquid nitrogen temperature, and features a Constant-PowerTM objective lens for optimal contrast/resolution balance.
 The Scios is a state-of-the-art DualBeam™ Scan Electron Microscopy (SEM) and Focused Ion Beam (FIB) system that allows for precision milling of samples. Technological advanced have increased throughput and milling precision, yielding higher quality results in less time, making it an ideal system for thinning of samples for subsequent TEM imaging.
The Scios is a state-of-the-art DualBeam™ Scan Electron Microscopy (SEM) and Focused Ion Beam (FIB) system that allows for precision milling of samples. Technological advanced have increased throughput and milling precision, yielding higher quality results in less time, making it an ideal system for thinning of samples for subsequent TEM imaging.
The Polara is a 300 KeV cryo-electron microscope from FEI. It is also equipped with a Gatan Bio-quantum K2 summit detector. This detector can be used for semi-automated data collection with SerialEM for both single particle and tomgraphy aplications. The Polara is operable at containment level 3.
 Let us introduce you to Jason Van Rooyen. Jason is the newest member of the ILO team and he is here to support our industrial users in the use of the state of the art facilities available at eBIC.
Let us introduce you to Jason Van Rooyen. Jason is the newest member of the ILO team and he is here to support our industrial users in the use of the state of the art facilities available at eBIC.
Jason is an experienced and enthusiastic structural biologist with many year’s experience in both cryo-EM and X-ray crystallography. He began his passion for EM as part of his Master’s degree and studies at the University of Cape Town. He then switched to protein crystallography for his PhD to solve the structure of a difficult protein for which EM was not up to the job at the time. He then did a post-doc at EMBL in Grenoble studying complexes and proteins with potential as drug targets against the pathogen Toxoplasma gondii using both EM and crystallography.
Following this, he supported the uptake of automated imaging techniques, and looked after acquisition, image processing, and data management for the EM unit in Cape Town. Jason is now back with his feet firmly planted into EM to join eBIC as an industry EM scientist where he will assist researchers and scientists from industry to leverage the state-of-the-art capabilities here at Diamond to provide novel and previously unattainable structural insights for the drug-discovery and biotech industries.
Would you like to know more about eBIC and how you can access it's facilities? Then please get in touch with Jason. He is able to offer you a variety of service options ranging from full service; a bespoke experimental design, data collection, data analysis and reporting service through to providing facilities for you to conduct your own experiments. Please do give him a call on 01235 778029 or send him an e-mail - jason.van-rooyen@diamond.ac.uk.
Diamond Light Source is the UK's national synchrotron science facility, located at the Harwell Science and Innovation Campus in Oxfordshire.
Copyright © 2022 Diamond Light Source
Diamond Light Source Ltd
Diamond House
Harwell Science & Innovation Campus
Didcot
Oxfordshire
OX11 0DE
Diamond Light Source® and the Diamond logo are registered trademarks of Diamond Light Source Ltd
Registered in England and Wales at Diamond House, Harwell Science and Innovation Campus, Didcot, Oxfordshire, OX11 0DE, United Kingdom. Company number: 4375679. VAT number: 287 461 957. Economic Operators Registration and Identification (EORI) number: GB287461957003.