___________________________________
Industrial Liaison Group:
Tel: +44 (0) 1235 778797
E-mail: [email protected]
Below you can see some of the practical applications of X-ray spectroscopy in case studies with our Industrial Liaison Users.

X-ray spectroscopy is a powerful tool for the determination of local atomic structure in materials that are not necessarily characterised by crystalline order. Spectroscopy can therefore be applied to materials whose constituents, such as atoms, molecules or ions, are not arranged in a highly ordered microscopic structure and do not form a crystal lattice that extends in all directions, creating a very powerful direct probe of chemical environments.
Atoms and molecules have unique spectra. These spectra can be used to detect, identify and quantify information about the atoms and molecules in a sample; in particular elemental composition, chemical state and physical properties of both inorganic material and biological systems. By sweeping through a range of photon energies and measuring the response, the absorption, reflectivity or fluorescence of the sample is measured.
Spectroscopy allows researchers to gain valuable information about the internal consituents of a sample and how they may change over varying conditions such as time, temperature or pressure.

It was 1666 when Newton showed that the white light from the sun could be dispersed into a continuous series of colours. Newton introduced the word "spectrum" to describe this phenomenon and it was this analysis of light that was the beginning of the science of spectroscopy.
In 1800, William Herschel demonstrated that the sun's radiation extended into the infrared, and in 1801 Johann Wilhelm Ritter made similar observations in the ultraviolet.
However, it was the achievements of Joseph Fraunhofer in the early 1800's that provided the quantitative basis for spectroscopy. Fraunhofer extended Newton's discovery by observing that the sun's spectrum, when sufficiently dispersed, was crossed by a large number of fine dark lines, now known as Fraunhofer lines.
Fraunhofer also developed the diffraction grating, an array of slits, which disperses light in much the same way as a glass prism, but with the advantage of being able to directly measure the wavelength of the diffracted beam. Earlier, Thomas Young (see our article on diffraction here) had demonstrated that a light beam passing through a slit emerges in a pattern of bright and dark fringes. Fraunhofer extended these studies to the case of two, three and many closely spaced slits, and the transmission grating was created. With this, he was able to directly measure the wavelengths of spectral lines.
Despite his enormous achievements, Fraunhofer did not understand the origin of the spectral lines he observed. It was not until 33 years after his death that Gustav Kirchhoff established that each element and compound has its own unique spectrum, and that by studying the spectrum of an unknown source, one could determine its chemical composition. With these advancements, spectroscopy became a true scientific discipline.
In the early 1800's many researchers studied spectra from sources such as flames, arcs and sparks. These sources were found to emit bright spectral lines, which were characteristic of the chemical elements in the flame. Leon Foucault, a French physicist, observed in 1848 that a flame containing sodium would absorb the yellow light emitted by a strong arc placed behind it. This was the first demonstration of a laboratory absorption spectrum.
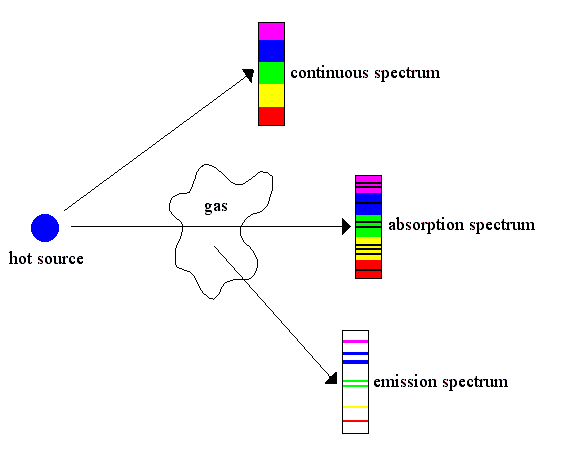 All of these facts were brought together in 1859 by Gustav Kirchhoff in his famous law, which states that the emitted power and absorbed power of light at a given wavelength are the same for all bodies at the same temperature. It follows that a gas, which radiates a line spectrum must, at the same temperature, absorb the spectral lines it radiates. From this, Kirchhoff and R. Bunsen explained that the Fraunhofer lines in the sun's spectrum were due to absorption of the continuous spectrum emitted from the hot interior of the sun by elements at the cooler surface.
All of these facts were brought together in 1859 by Gustav Kirchhoff in his famous law, which states that the emitted power and absorbed power of light at a given wavelength are the same for all bodies at the same temperature. It follows that a gas, which radiates a line spectrum must, at the same temperature, absorb the spectral lines it radiates. From this, Kirchhoff and R. Bunsen explained that the Fraunhofer lines in the sun's spectrum were due to absorption of the continuous spectrum emitted from the hot interior of the sun by elements at the cooler surface.
By recognising that each atom and molecule has its own characteristic spectrum, Kirchhoff and Bunsen established spectroscopy as a scientific tool for probing atomic and molecular structure, and founded the field of spectrochemical analysis for analysing the composition of materials.
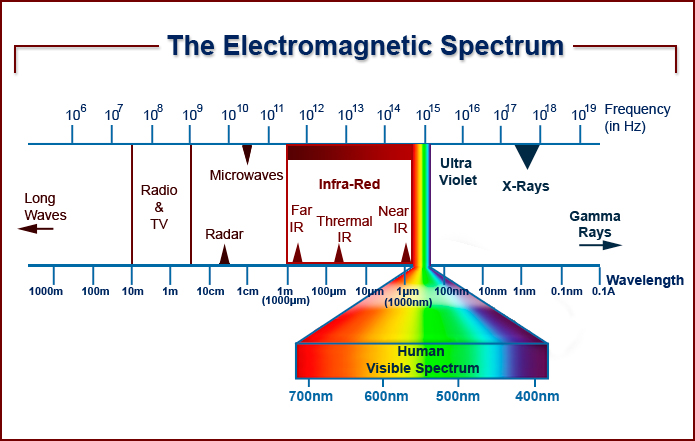
When a beam of white light strikes a triangular prism it is separated into its various components (ROYGBIV). This is known as a spectrum.
The electromagnetic spectrum extends from below the low frequencies used for modern radio communication to gamma radiation at the short-wavelength (high-frequency) end, thereby covering wavelengths from thousands of kilometers down to a fraction of the size of an atom. Visible light lies toward the shorter end, with wavelengths from 400 to 700 nanometres.
Nearly all types of electromagnetic radiation can be used for spectroscopy to study and characterise matter. When using X-rays the photoelectric effect occurs.
The photoelectric effect refers to the emission, or ejection, of electrons from the surface of a sample in response to electromagnetic radiation, or light. Energy contained within the electromagnetic radiation is absorbed by electrons within the sample, giving the electrons sufficient energy to be 'knocked' out of, that is, emitted from, the sample.
In the X-ray region atoms absorb X-rays sharply at certain wavelengths (called absorption edges) that are characteristic of that particular atomic species. So if you shine a particular frequency, or energy, of X-rays onto a sample, you can control the type of electron that is emitted.
The process occurs as follows:
1. The core level electron absorbs the energy from the X-ray;
2. If sufficient energy is provided, the electron is ejected from the atom forming a photoelectron which affects neighbouring atoms;
3. The atom is left in excited state with an empty electronic level i.e. core hole.
An absorption graph is produced which provides researchers with element-specific information from the sample. This can include which elements are present, where they are located, the density of the element, what are the neighbouring atoms and the distance between those atoms.
At Diamond, we employ three main techniques in the field of spectroscopy:
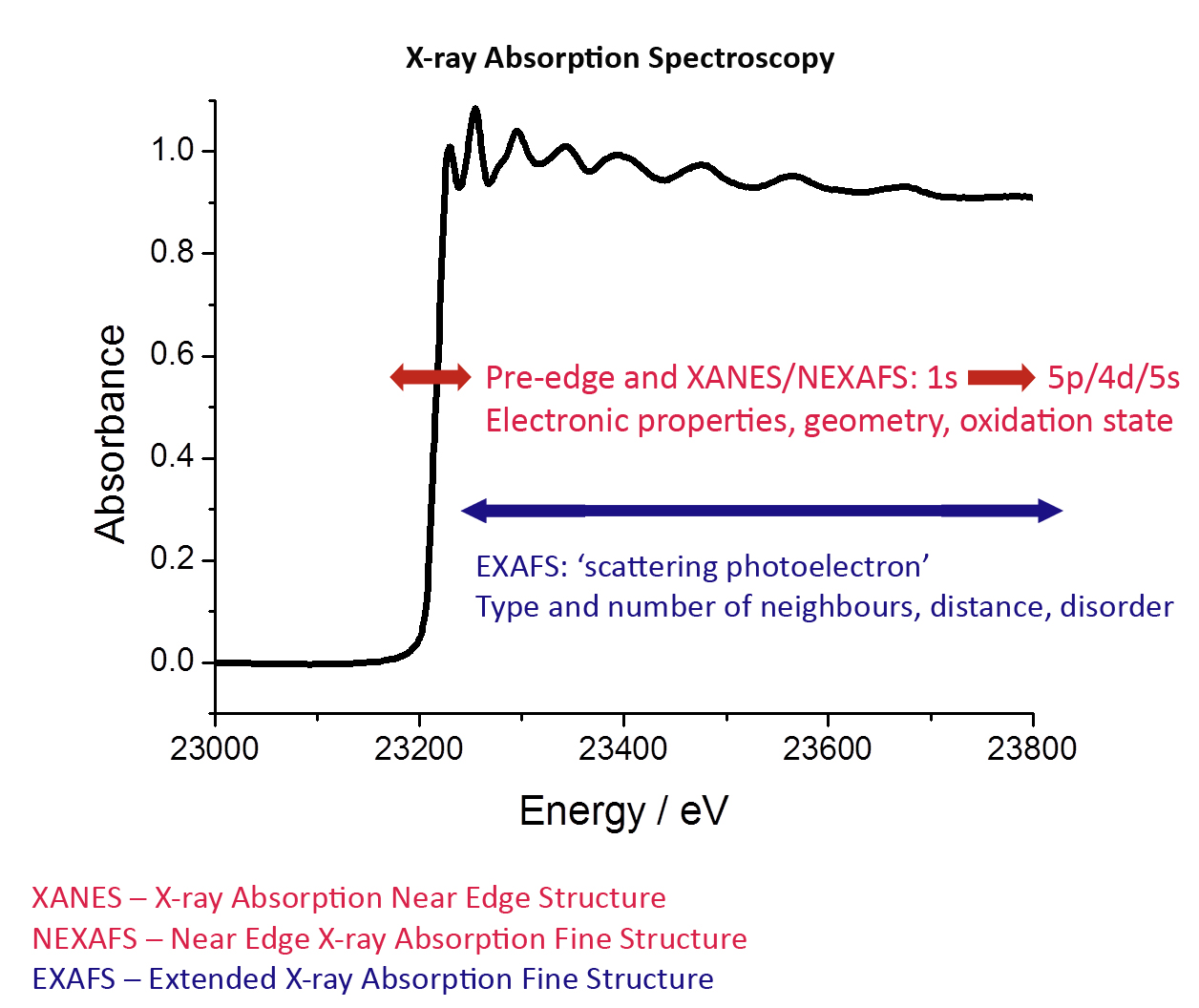
X-ray absorption spectroscopy (XAS) is an element-specific technique dedicated to study electronic and structural properties of selected element. This unique technique is greatly appreciated when studying non-crystalline materials.
At characteristic wavelengths the X-ray absorption of an element changes dramatically, these are called absorption edges. XAS is a technique that can be divided into XANES - near the absorption edge, the spectra may contain fine structure that reveals the electronic and geometrical environment of the absorbing atom - and further from the edge EXAFS reveals the atomic environment local to the element. Both can be used to follow reactions on timescales down to the millisecond.
Benefits of Synchrotron Techniques
Useful for

X-ray absorption spectroscopy is important in gaining structural understanding of a range of materials, including catalysts, biomaterials, novel materials with special electronic properties such as superconductivity, dilute species in fluids, and complex inhomogeneous materials.
It can provide information on bio-remediation processes, study minute minerals returned from space missions and be used to understand chemical reactions such as heterogeneous catalysis and hydrothermal synthesis of industrial materials.
Beamlines
B18 - I08 - I18 - I20 - B07- I14 - I06
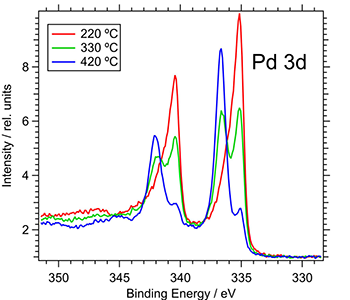 X-ray photoelectron spectroscopy (XPS) is a surface sensitive technique used to determine the elemental composition, chemical and electronic state of the elements within a studied material. XPS spectra are acquired by irradiating a solid surface with X-rays while simultaneously measuring the kinetic electrons that are discharged from the top ~100 Å (10 nm) of a sample being measured.
X-ray photoelectron spectroscopy (XPS) is a surface sensitive technique used to determine the elemental composition, chemical and electronic state of the elements within a studied material. XPS spectra are acquired by irradiating a solid surface with X-rays while simultaneously measuring the kinetic electrons that are discharged from the top ~100 Å (10 nm) of a sample being measured.
Benefits of synchrotron techniques
Useful for
 XPS using a synchrotron enables users to study the interfacial structures and composition of thin films and nanoparticulates, and the doping interactions in electrolyte materials for fuel cell applications. Perfect for performing in situ characterisation, XPS can be used to study the surface composition of heterogeneous catalysts under working conditions, study biological and pharmaceutical samples under equilibrium water-vapour conditions, and directly undertake spectroscopy of surfaces of liquids which offers insight into the atmospheric chemistry of aerosols.
XPS using a synchrotron enables users to study the interfacial structures and composition of thin films and nanoparticulates, and the doping interactions in electrolyte materials for fuel cell applications. Perfect for performing in situ characterisation, XPS can be used to study the surface composition of heterogeneous catalysts under working conditions, study biological and pharmaceutical samples under equilibrium water-vapour conditions, and directly undertake spectroscopy of surfaces of liquids which offers insight into the atmospheric chemistry of aerosols.
Beamlines
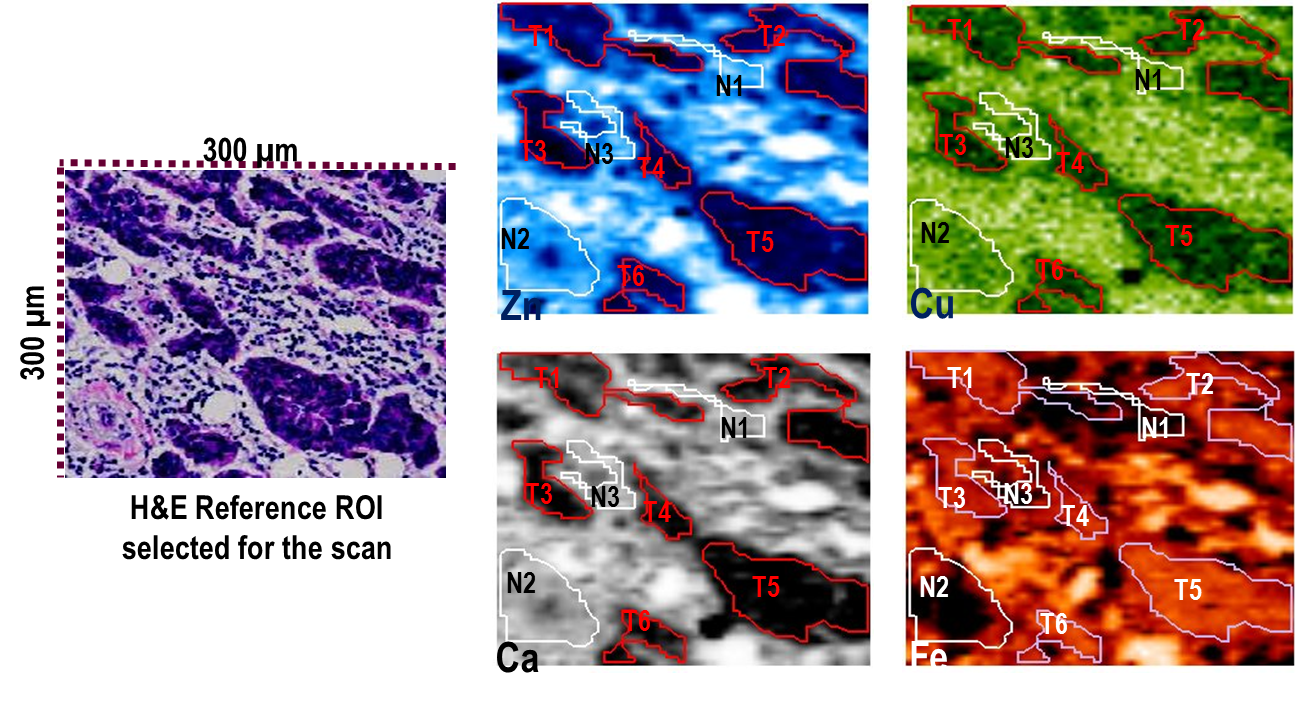
X-ray Fluorescence (XRF) occurs when the inner shell electrons of atoms in the sample get excited by the X-rays and subsequently release X-ray photons when the system relaxes, that is when the electrons transition from the higher energy levels of the atom to the vacant inner shell.
The beauty of this process is that each secondary X-ray photon (sometimes called characteristic radiation) emitted from the sample has a specific energy which is a fingerprint of the atom from which it has originated. By measuring the energy of the secondary photons it is possible to establish the elemental composition of the sample at the point where the X-ray beam hits the sample. Typically a special type of detector known as an energy-dispersive detector is used to precisely measure the energy of each photon. The plot of the number of photon counts versus their energy, the X-ray spectrum, typically shows a number of peaks which are directly associated with specific elements, so by just glancing at the spectrum it is possible to quickly deduce which elements are present in the sample.
Benefits of synchrotron techniques
Useful for
 X-ray fluorescence spectroscopy can be used to study the chemical composition of virtually anything and is becoming an advanced and essential analytical technique in life and environmental sciences, medical applications, archaeological and cultural heritage applications, forensic chemistry, industrial applications, and earth and planetary sciences. Some of the research taking place at synchrotrons has helped scientists to study historical artefacts, develop highly nutritious food, and investigate Alzheimer’s disease.
X-ray fluorescence spectroscopy can be used to study the chemical composition of virtually anything and is becoming an advanced and essential analytical technique in life and environmental sciences, medical applications, archaeological and cultural heritage applications, forensic chemistry, industrial applications, and earth and planetary sciences. Some of the research taking place at synchrotrons has helped scientists to study historical artefacts, develop highly nutritious food, and investigate Alzheimer’s disease.
Beamlines
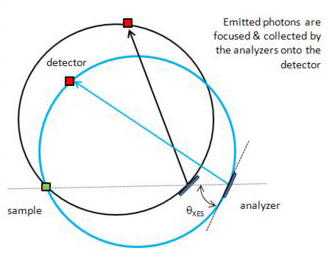

X-ray emission spectroscopy (XES) is one of the so-called "photon-in - photon-out" spectroscopies in which a core electron is excited by an incident X-ray photon and then this excited state decays by emitting an X-ray photon to fill the core hole. The energy of the emitted photon is the energy difference between the involved electronic levels. The analysis of the energy dependence of the emitted photons is the aim of the X-ray emission spectroscopy.
Benefits of synchrotron techniques
Useful for

Catalysts - direct studies of the structure and interactions of catalysts with chemical reagents under rapidly changing environmental conditions – three-way catalysts, fuel cells, photochemical processes and solution chemistry.
Environmental - metal speciation of toxic materials to handle the remediation of environmental contamination, processes used for the disposal of toxic materials, studying rocks, soils, sediments, plant materials, pollutants and radioactive waste issues in climate change.
Material Science - samples under realistic conditions of high pressures and temperatures, kinetic processes in operating electrochemical cells, design and characterisation of novel, advanced materials.
Biology - determination of the structure of metalloproteins, studying biochemical processes such as the life mechanisms of photosynthesis or respiration.
Beamlines
Would you like to know more about X-ray spectroscopy and how you can apply it to your research? Do you perhaps have a structural problem that you are unable to solve in your lab or a material you wish to find out more about? Then please get in touch with the Industrial Liaison Team at Diamond.
The Industrial Liaison team at Diamond is a group of professional, experienced scientists with a diverse range of expertise, dedicated to helping scientists and researchers from industry access the facilities at Diamond.
We’re all specialists in different techniques and have a diverse range of backgrounds so we’re able to provide a multi-disciplinary approach to solving your research problems.
We offer services ranging from full service; a bespoke experimental design, data collection, data analysis and reporting service through to providing facilities for you to conduct your own experiments. We’re always happy to discuss any enquiries or talk about ways in which access to Diamond’s facilities may be beneficial to your business so please do complete an enquiry form or give us a call on 01235 778797. You can keep in touch with the latest development by following us on Twitter @DiamondILO or LinkedIn

Diamond Light Source is the UK's national synchrotron science facility, located at the Harwell Science and Innovation Campus in Oxfordshire.
Copyright © 2022 Diamond Light Source
Diamond Light Source Ltd
Diamond House
Harwell Science & Innovation Campus
Didcot
Oxfordshire
OX11 0DE
Diamond Light Source® and the Diamond logo are registered trademarks of Diamond Light Source Ltd
Registered in England and Wales at Diamond House, Harwell Science and Innovation Campus, Didcot, Oxfordshire, OX11 0DE, United Kingdom. Company number: 4375679. VAT number: 287 461 957. Economic Operators Registration and Identification (EORI) number: GB287461957003.