____________________________________
Industrial Liaison Group:
Tel: +44 (0) 1235 778797
E-mail: [email protected]
In today’s battle against climate change, replacement of conventional cars with battery electric vehicles (BEV) offers an opportunity to significantly reduce future carbon dioxide emissions. Currently, BEVs employ lithium-ion batteries which are expensive to produce and have significant drawbacks; their safety and limited transport being the main issues. Significant interest is therefore being shown in replacing these lithium-ion batteries with a lithium-sulfur battery (LiS or Li2S/Si), which operates using a cheap and abundant raw material with about a two-fold higher specific energy compared to lithium-ion batteries.
The significant theoretical advantages of lithium-sulfur batteries over lithium-ion technology have generated a lot of interest in the system, but the development of practical prototypes, which could be successfully incorporated into BEVs, remains slow, with issues such as safety and deactivation. One of the main barriers to achieving such breakthroughs is the lack of fundamental understanding of the mechanism behind the operation of Li-S batteries. In particular, it is not yet clear what the charge and discharge mechanisms are, if the formed polysulfide species are reversible or not, and how all these processes depend on type and amount of electrolyte and the amount of active material. Consequently, there is a pressing need for performing operando characterisation of Li-S batteries under a variety of conditions to identify fundamental aspects of the charging, discharging and deactivation processes.
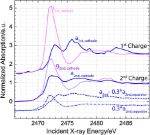
Researchers from Germany and Amsterdam performed sulfur K-edge X-ray absorption spectroscopy (XAS) measurements on the I18 beamline at Diamond Light Source. Spatially resolved XAS with a small X-ray beam in the specially designed cell, allowed experimentation at different positions in the battery cell, to unravel electrode- vs. electrolyte-only processes.
This work highlights the advantages of spatially resolved XAS characterisation, which can detect solution phase polysulfides and amorphous as well as crystalline solids, while also differentiating between the species forming in the electrode and the separator. Using this technique, the researchers were able to obtain information about intermediates present in the cathode and the separator of an operating Li-S battery during discharging and charging. These identified pathways are expected to assist in reaching higher storage capacities in Li-S batteries and avoid deactivation by guiding both the development of models of Li-S battery operation and the design of improved cathode structures and electrolyte components.
.jpg)


XAS is the only technique, which allows the detailed structural and electronic characterisation of solid and liquid species under operando conditions. Being able to do this as a function of position in the cell, we were able to obtain new and unique information, which allows further design and optimisation in a rational manner.
Prof Dr Moniek Tromp, University of Amsterdam

Diamond Light Source is the UK's national synchrotron science facility, located at the Harwell Science and Innovation Campus in Oxfordshire.
Copyright © 2022 Diamond Light Source
Diamond Light Source Ltd
Diamond House
Harwell Science & Innovation Campus
Didcot
Oxfordshire
OX11 0DE
Diamond Light Source® and the Diamond logo are registered trademarks of Diamond Light Source Ltd
Registered in England and Wales at Diamond House, Harwell Science and Innovation Campus, Didcot, Oxfordshire, OX11 0DE, United Kingdom. Company number: 4375679. VAT number: 287 461 957. Economic Operators Registration and Identification (EORI) number: GB287461957003.