___________________________________
Industrial Liaison Group:
Tel: +44 (0) 1235 778797
E-mail: industry@diamond.ac.uk

Powder diffraction is an important technique for characterising the crystallographic structure of materials.
The technique allows systems with single or multiple crystalline phases to be studied and can be used to identify the constituent phases within a material, follow phase transitions and perform full crystallographic structural determination of unknown materials.
Powder diffraction is a popular and powerful tool for studying a great variety of systems, from naturally occurring materials and minerals such as calcium carbonate, to novel man made pharmaceutical medicines, and determining the strain within engineering components.

The route to the discovery of the powder diffraction (PD) method was not straightforward. Some results of experiments performed on powders or samples having a polycrystalline component were published as early as 1913 by W. Friedrich and H.B. Keene, but the meaning of the observed rings was not understood at that time.
The first experiments on powders, or polycrystals, were by Paul Scherrer, a Swiss physicist, and were guided by Peter Debye's suggestion that "specific diffraction effects should be produced with X-rays by the regular spacing of electrons on circular orbits". The experiments were initially unsuccessful but this did not discourage Debye and Scherrer: they constructed a camera (now known as a Debye-Scherrer camera) which, when joined with a source of characteristic X-rays, produced a diffraction pattern with unexpectedly sharp lines. Explanation of this pattern became the basis of the powder diffraction method we use today.
 Across the water, Albert W. Hull, who until 1916 had nothing in common with crystallography, became interested in the magnetic properties of some elements, particularly iron. After a visit from Sir William Bragg to his laboratory talking about the X-ray crystal analysis work which he and his son were doing, Hull approached Bragg and asked if he had found the crystal structure of iron. The response given was ‘no, we have tried but haven’t succeeded’ and that became the challenge for Hull; to find the crystal structure of iron. He constructed a camera and collected data but unfortunately his assistant made a calculation error and found the powder diffraction patterns did not fit the Braggs' values. So Hull busied himself with solving the structure of tungsten powder and of Fe-Si single crystal, and only at a later date started new calculations for polycrystalline iron data.
Across the water, Albert W. Hull, who until 1916 had nothing in common with crystallography, became interested in the magnetic properties of some elements, particularly iron. After a visit from Sir William Bragg to his laboratory talking about the X-ray crystal analysis work which he and his son were doing, Hull approached Bragg and asked if he had found the crystal structure of iron. The response given was ‘no, we have tried but haven’t succeeded’ and that became the challenge for Hull; to find the crystal structure of iron. He constructed a camera and collected data but unfortunately his assistant made a calculation error and found the powder diffraction patterns did not fit the Braggs' values. So Hull busied himself with solving the structure of tungsten powder and of Fe-Si single crystal, and only at a later date started new calculations for polycrystalline iron data.
After the Second World War, rapid progress in powder diffraction methods took place. The progress in the speed of data collection and quality of results was due to the development of instrumentation such as automated diffractometers, goniometers, generators and detectors, as well as the availability of digitised data and to numerical methods of pattern solving and refinement. Ultimately, classical X-ray diffraction in home laboratories suffers from limitations connected with restricted wavelength selection and slow data collection and these difficulties were not overcome until the introduction of synchrotron X-ray sources.
During the 1970s dedicated synchrotron laboratories began operation and the powder diffraction results from such sources have made a significant contribution to the scientific world.
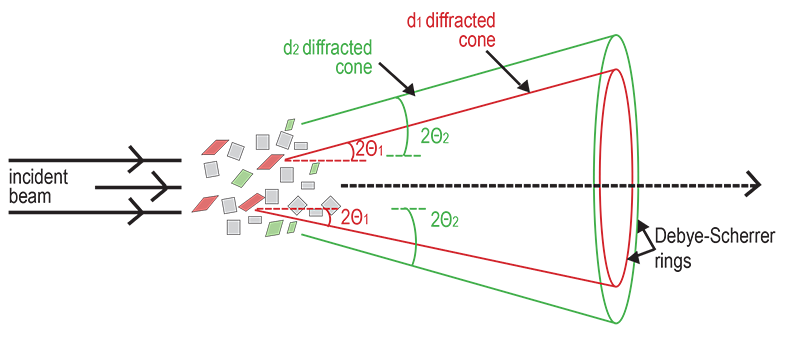
When an X-ray is focused on a crystal, it diffracts in a pattern characteristic of the crystal structure. In powder X-ray diffraction (XRD), the diffraction pattern for the bulk material of a polycrystalline solid is obtained, rather than of a single crystal, which doesn't necessarily represent the overall material. A diffraction pattern plots intensity against the angle of the detector, 2θ.
X-ray diffraction is based on constructive interference of monochromatic X-rays and a crystalline sample. X-rays are generated, filtered to produce monochromatic radiation, collimated to concentrate, and directed toward the sample. When the X-rays reach the sample, the atoms of the sample act just like a diffraction grating, producing bright spots at particular angles. By measuring the angle where these bright spots occur, the spacing of the diffraction grating can be determined by Bragg's law. Because the sample itself is the diffraction grating, this spacing is the atomic spacing.
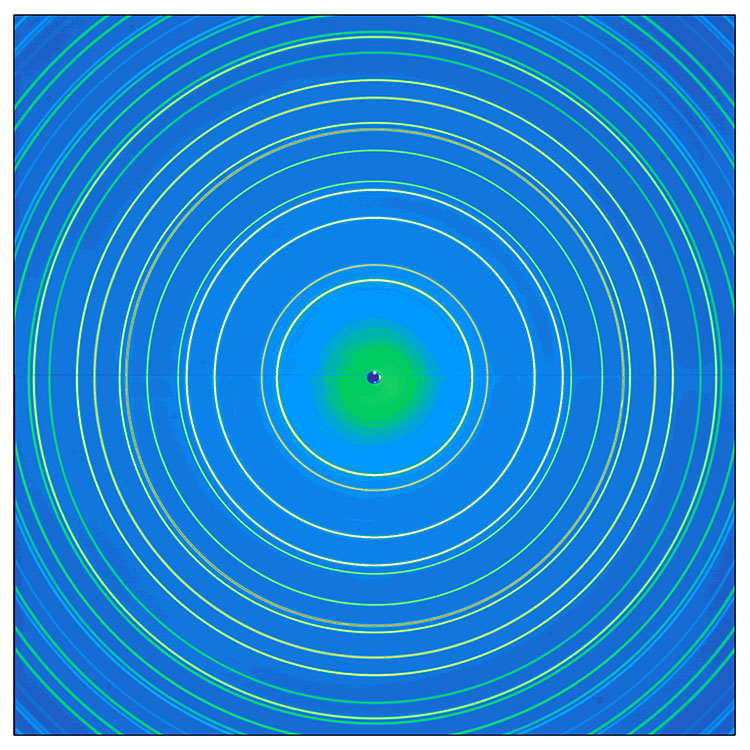
In powder diffraction, every possible crystalline orientation is represented equally in a powdered sample, and powder X-ray diffraction operates under the assumption that the sample is randomly arranged. Therefore, a statistically significant number of each of the planes of the crystal structure will be in the proper orientation to diffract the X-rays. Therefore, each plane will be represented in the signal. In practice, it is sometimes necessary to rotate the sample orientation to eliminate the effects of texturing and achieve true randomness. These diffracted X-rays are then detected, processed and counted. By scanning the sample through a range of 2θ angles, all possible diffraction directions of the sample can be gathered due to the random orientation of the powdered material. This will produce cones of diffracted rays which intersect a flat photographic plate as circles, known as Debye-Scherrer rings, named after the early powder diffraction camera invented by Debye, Scherrer and Hull.
 Materials are made of atoms. Knowledge of how atoms are arranged into crystal structures and microstructures is the foundation on which we build our understanding of the synthesis, structure and properties of materials.
Materials are made of atoms. Knowledge of how atoms are arranged into crystal structures and microstructures is the foundation on which we build our understanding of the synthesis, structure and properties of materials.
X-ray powder diffraction is one of the primary techniques used to characterise materials, providing structural information even when the crystallite size is too small for single crystal X-ray diffraction methods. It is most widely used for the identification of unknown crystalline materials (e.g. minerals, inorganic compounds). Determination of unknown solids is critical to studies in geology, environmental science, material science, engineering and biology.
Other applications include:
With specialised techniques, X-ray powder diffraction can be also be used to:
There are three beamlines at Diamond Light Source that can carry out powder diffraction experiments; I11, I12 and I15.
 High-Resolution and Time-Resolved Experiments
High-Resolution and Time-Resolved Experiments
The combination of very high angular resolution, high count rates and controlled environmental conditions on I11 make it possible to carry out detailed structural analysis of complex materials. These studies are important to many fundamental areas of the physical, life and environmental sciences as well as pharmaceutical, engineering and industrial materials.
This versatile beamline has the resolving power to probe deep into sample structures ( Δ d/d ~ 10-5-6), to detect rapid changes ( Δ t ~ ms-s) under non-ambient conditions as they occur and to perform resonant diffraction in order to solve complex structures containing low normal electron contrasts.
Long Duration Experiments
This new facility is unique within the synchrotron world and is used for scientific investigations where samples will be maintained at, or cycled through, various non-ambient conditions for extended periods.
To complement the existing I11 high resolution and fast time-resolved facilities, the LDE facility will open up new opportunities for those experiments which require weeks to months of periodically monitoring “slow” changes. It will be of particular benefit to certain research areas such as batteries and fuel cells where important information on the development of phases over time cannot be obtained via ex situ methods. Other research areas that will similarly benefit include studies of crystallisation, nuclear waste storage materials, gas storage, mineral evolution, seasonal effects, thermal and electrical power cycling and corrosion science, etc.
 Beamline I12-JEEP (Joint Engineering, Environmental, and Processing) is a high energy X-ray beamline for imaging, diffraction and scattering, which operates at energies of 53-150 keV.
Beamline I12-JEEP (Joint Engineering, Environmental, and Processing) is a high energy X-ray beamline for imaging, diffraction and scattering, which operates at energies of 53-150 keV.
I12 is suitable for experiments, which utilise high-energy X-rays, time-resolved in situ measurements, monochromatic or polychromatic (“white beam”) X-rays, imaging and diffraction techniques, and complex sample environments.
At I12 you can perform a range of experimental techniques which include static and time-resolved powder diffraction using monochromatic radiation and time-resolved powder diffraction using energy-dispersive radiation.
 The Extreme Conditions beamline I15 is dedicated to X-ray powder and single-crystal diffraction under extreme pressures and temperatures but can be used for a wide range of different sample types.
The Extreme Conditions beamline I15 is dedicated to X-ray powder and single-crystal diffraction under extreme pressures and temperatures but can be used for a wide range of different sample types.
The beamline provides monochromatic high-energy X-rays from 20 to 80 keV. These X-rays focussed and collimated down to <20 μm can penetrate into complex sample assemblies permitting detailed mapping of structural order or disorder, chemical fingerprinting, or single crystal structure determination.
I15 is a multi-disciplinary beamline with research topics including fundamental physics and chemistry, earth and planetary science, material science, and also bio-molecular science.
Would you like to know more about X-ray powder diffraction and how you can apply it to your research? Do you perhaps have a structural problem that you are unable to solve in your lab or a material you wish to find out more about? Then please get in touch with the Industrial Liaison Team at Diamond.
The Industrial Liaison team at Diamond is a group of professional, experienced scientists with a diverse range of expertise, dedicated to helping scientists and researchers from industry access the facilities at Diamond. We’re all specialists in different techniques and have a diverse range of backgrounds so we’re able to provide a multi-disciplinary approach to solving your research problems. We offer services ranging from full service; a bespoke experimental design, data collection, data analysis and reporting service through to providing facilities for you to conduct your own experiments.
We’re always happy to discuss any enquiries or talk about ways in which access to Diamond’s facilities may be beneficial to your business so please do give us a call on 01235 778797 or send us an e-mail. You can keep in touch with the latest development by following us on Twitter @DiamondILO orLinkedIn.
Diamond Light Source is the UK's national synchrotron science facility, located at the Harwell Science and Innovation Campus in Oxfordshire.
Copyright © 2022 Diamond Light Source
Diamond Light Source Ltd
Diamond House
Harwell Science & Innovation Campus
Didcot
Oxfordshire
OX11 0DE
Diamond Light Source® and the Diamond logo are registered trademarks of Diamond Light Source Ltd
Registered in England and Wales at Diamond House, Harwell Science and Innovation Campus, Didcot, Oxfordshire, OX11 0DE, United Kingdom. Company number: 4375679. VAT number: 287 461 957. Economic Operators Registration and Identification (EORI) number: GB287461957003.