Find out more about our ambitious upgrade project, delivering more brightness, more coherence, and greater speed of analysis to UK science. More about Diamond-II
![]()
Find out more about Diamond's response to virus research.
![]()
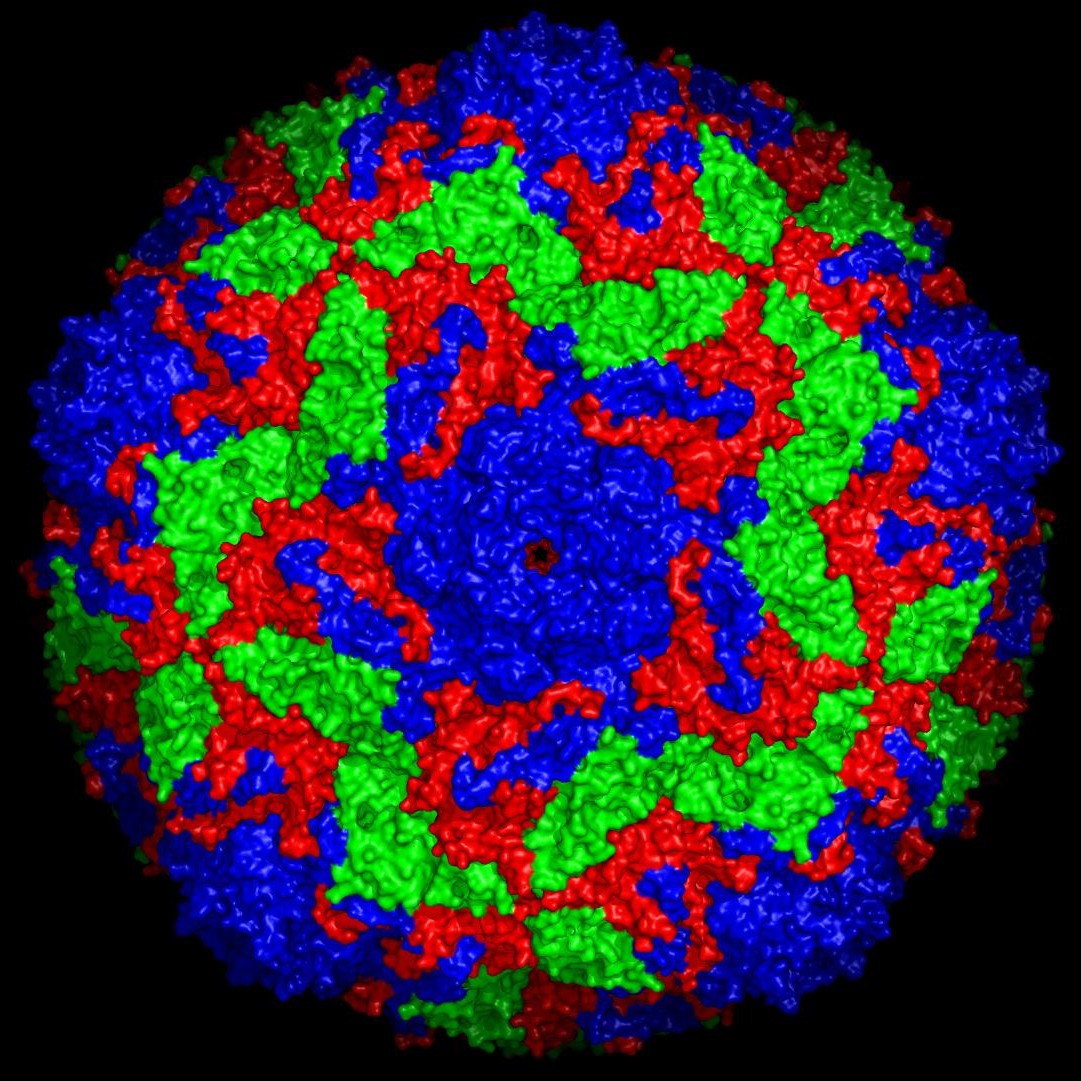
An international team of scientists, including contributors from Diamond Light Source, has for the first time used an X-ray free-electron laser to unravel a virus structure on the atomic level. They imaged the bovine enterovirus 2 (BEV2) virus, which can cause miscarriages, stillbirths, and infertility in cattle.
The method used dramatically reduces the amount of virus material required, while also allowing the investigations to be carried out several times faster than before, report the researchers in Nature Methods.
X-ray crystallography is one of the most prolific tools used by structural biologists at facilities like Diamond to determine the structure of thousands of biological materials. However the crystals are often difficult to grow, remaining tiny, and easily damaged by X-rays.
“X-ray free-electron (XFEL) lasers have opened up a new path to protein crystallography,” explains Dr Armin Wagner, Principal Beamline Scientist on Diamond’s I23 beamline, and co-author on the paper. “Their extremely intense pulses can be used to analyse even extremely tiny crystals that would not produce a sufficiently bright diffraction image using other X-ray sources.”
Because of the high intensity and short duration (typically 50 fs) of the X-ray pulse created by an XFEL, each of the microcrystals analysed can only produce a single diffraction image before the X-ray pulse destroys them. To complete a structural analysis, hundreds, or very often tens of thousands of diffraction images are required.
Consequently, fresh samples must be delivered to the interaction region, preferably at the same frequency as the XFEL to limit sample waste. Several such serial femtosecond crystallography (SFX) methods are available to present crystals to the XFEL beam and already more than 150 structures have been solved using SFX strategies. However, most methods have either to slow down the XFEL pulse rate significantly or require large amounts of sample as many crystals are missed.
“Each time a pulse from the XFEL happens to strike a microcrystal, a diffraction image is produced,” adds Dr Alke Meents, from DESY, Hamburg and principal investigator on the paper. “However the hit rate is typically less than two percent, so most of the precious microcrystals end up unused in the sample collection container.”
In order to use the beamtime and sample material more efficiently, the team developed a new method. The scientists use a micro-patterned chip containing thousands of tiny pores to hold the protein crystals. The X-ray laser then scans the chip line by line, synchronising the positioning of the pores to the full XFEL pulse repetition rate of 120 Hz.
The project, led by DESY scientists, involved researchers from the University of Oxford, the University of Eastern Finland, the Swiss Paul Scherrer Institute, the Lawrence Berkeley National Laboratory in the US and the US SLAC National Accelerator Laboratory. Diamond scientists have collaborated with the team at DESY, with much of the development and testing of the micro-patterned chip being performed on Diamond’s I02 and I24 beamlines.
As a world-leading facility for protein crystallography Diamond is also the base for the UK XFEL Hub, which aims to help scientists to prepare for their experiments at XFEL facilities. UK funders have also contributed to the construction of the European XFEL.
“Applications for beamtime at XFEL facilities around the globe are incredibly competitive, and therefore the beamtime is really precious to the users” explains Dr Allen Orville, Principal Scientist of the UK XFEL Hub. “By offering comprehensive testing of new sample tools and triggers at Diamond, we’re able to make sure they can focus on research during the XFEL beamtime, as the experimental setup will be optimised.”
“One other example of our work is tools that exploit triggering and synchronisation strategies to deliver crystal droplets directly into the pulse using focused sound waves,” he continues.
“The UK XFEL Hub is a fine example of how the UK is taking a leading role in the development of global new facilities, by developing new techniques and expertise,” adds Professor Andrew Harrison, CEO at Diamond. “We are pleased that the UK XFEL Hub is already having an impact upon the technology used for our beamlines, as well as the cryo-electron microscopes at our electron Bio-Imaging Centre (eBIC).”
Find out more about his story on the DESY website.
P. Roedig, H. M. Ginn, T. Pakendorf, G. Sutton, K. Harlos, T. S. Walter, J. Meyer, P. Fischer, R. Duman, I. Vartiainen, B. Reime, M. Warmer, A. S. Brewster, I. D. Young, T. Michels-Clark, N. K. Sauter, M. Sikorsky, S. Nelson, D. S. Damiani, R. Alonso-Mori, J. Ren, E. E. Fry, C. David, D. I. Stuart, A. Wagner, A. Meents High-speed fixed-target serial virus crystallography Nature Methods, 2017; DOI: 10.1038/nmeth.4335
P. Roedig, I. Vartiainen, R. Duman, S. Panneerselvam, N. Struebe, O. Lorbeer, M. Warmer, G. Sutton, D. I. Stuart, E. Weckert, C. David, A. Wagner, A. Meents A micro-patterned silicon chip as sample holder for macromolecular crystallography experiments with minimal background scattering Sci. Rep. 5 (2015) 10451.
P. Roedig, R. Duman, J. Sanchez-Weatherby, I. Vartiainen, A. Burkhardt, M. Warmer, C. David, A. Wagner, A. Meents Room-temperature macromolecular crystallography using a micro-patterned silicon chip with minimal background J. Appl. Cryst. 49 (2016) 968-975.
Diamond Light Source is the UK's national synchrotron science facility, located at the Harwell Science and Innovation Campus in Oxfordshire.
Copyright © 2022 Diamond Light Source
Diamond Light Source Ltd
Diamond House
Harwell Science & Innovation Campus
Didcot
Oxfordshire
OX11 0DE
Diamond Light Source® and the Diamond logo are registered trademarks of Diamond Light Source Ltd
Registered in England and Wales at Diamond House, Harwell Science and Innovation Campus, Didcot, Oxfordshire, OX11 0DE, United Kingdom. Company number: 4375679. VAT number: 287 461 957. Economic Operators Registration and Identification (EORI) number: GB287461957003.