Keep up to date with the latest research and developments from Diamond. Sign up for news on our scientific output, facility updates and plans for the future.
David Hall, Village Coordinator
Diamond’s Macromolecular Crystallography (MX) Village provides a broad range of beamlines with specialised capabilities in determining the three dimensional structures of large biological molecules. MX is a vital tool for linking structure to function, and for investigating protein folding and other structural information. The challenges today also lie in understanding the dynamics of protein function within complex assemblies and cellular pathways.
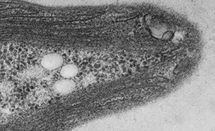 Methane is a more potent greenhouse gas than carbon dioxide, and atmospheric levels are increasing. Bacteria called methanotrophs can oxidise methane, using an enzyme called particulate methane monooxygenase (pMMO). pMMO requires copper in order to oxidise methane, and there are abundant pools of soluble copper within methanotrophs. Identifying the proteins that can bind copper in methanotrophs is important for potential uses of these organisms in reducing atmospheric methane, and in the industrial production of bioenergy, bulk, and fine chemicals. Read More
Methane is a more potent greenhouse gas than carbon dioxide, and atmospheric levels are increasing. Bacteria called methanotrophs can oxidise methane, using an enzyme called particulate methane monooxygenase (pMMO). pMMO requires copper in order to oxidise methane, and there are abundant pools of soluble copper within methanotrophs. Identifying the proteins that can bind copper in methanotrophs is important for potential uses of these organisms in reducing atmospheric methane, and in the industrial production of bioenergy, bulk, and fine chemicals. Read More
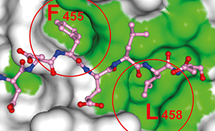 Autophagy is an important waste-disposal function in human cells, identifying and removing disease organisms and damaged organelles - cell structures with specific functions, such as mitochondoria, nucleus, and peroxisome. The exact process by which damaged or mutated organelles are identified and then selected for removal is not yet understood. Read More
Autophagy is an important waste-disposal function in human cells, identifying and removing disease organisms and damaged organelles - cell structures with specific functions, such as mitochondoria, nucleus, and peroxisome. The exact process by which damaged or mutated organelles are identified and then selected for removal is not yet understood. Read More
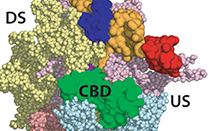 RNA polymerase (RNAP) is a fundamental piece of cellular machinery required for making messenger RNA from DNA in all cell types. Despite the large amount of structural information that has been obtained over the last 15 years, the means by which RNAP is regulated is still not completely understood. In bacteria, the RNAP is assisted by proteins called sigma factors, which recruit RNAP to specific regions on the DNA called promoters. One such sigma factor, known as sigma54 (σ54), recruits RNAP during special circumstances like stress and infection and inhibits transcription until additional activators intervene. Read More
RNA polymerase (RNAP) is a fundamental piece of cellular machinery required for making messenger RNA from DNA in all cell types. Despite the large amount of structural information that has been obtained over the last 15 years, the means by which RNAP is regulated is still not completely understood. In bacteria, the RNAP is assisted by proteins called sigma factors, which recruit RNAP to specific regions on the DNA called promoters. One such sigma factor, known as sigma54 (σ54), recruits RNAP during special circumstances like stress and infection and inhibits transcription until additional activators intervene. Read More
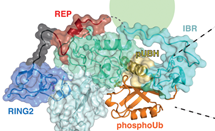 Parkinson’s disease affects millions of people worldwide, with the sporadic form typically occurring in people aged over 60 years. However, inherited versions of the disease can emerge in individuals who are in their twenties. Early-onset Parkinson’s disease is genetically linked to the proteins Parkin and PINK1, which perform crucial quality-control functions in the cell, and cause the condition when mutated. Read More
Parkinson’s disease affects millions of people worldwide, with the sporadic form typically occurring in people aged over 60 years. However, inherited versions of the disease can emerge in individuals who are in their twenties. Early-onset Parkinson’s disease is genetically linked to the proteins Parkin and PINK1, which perform crucial quality-control functions in the cell, and cause the condition when mutated. Read More
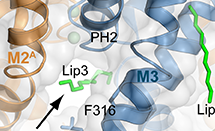 Treatments for cardiovascular disease, neurological disorders, and pain can be developed by understanding the flow of ions across cell membranes. Potassium 2-pore domain (K2P) channels are important clinical targets, but the mechanisms that control how these channels open and close were not well understood. TREK-2 is an archetypal K2P channel regulated by a range of diverse stimuli, including mechanical changes to the membrane, chemical changes such as pH, biological molecules and many pharmacological agents such as volatile anaesthetics, neuroprotective drugs, and antidepressants such as fluoxetine (Prozac). Read More
Treatments for cardiovascular disease, neurological disorders, and pain can be developed by understanding the flow of ions across cell membranes. Potassium 2-pore domain (K2P) channels are important clinical targets, but the mechanisms that control how these channels open and close were not well understood. TREK-2 is an archetypal K2P channel regulated by a range of diverse stimuli, including mechanical changes to the membrane, chemical changes such as pH, biological molecules and many pharmacological agents such as volatile anaesthetics, neuroprotective drugs, and antidepressants such as fluoxetine (Prozac). Read More
Beamline I24, the tuneable microfocus MX beamline, has recently completed its end station upgrade, a project that necessitated a complete rebuild of the beamline. Users can now exploit improved sample viewing, new sample positioning systems, and a novel dual goniometer design. The upgrade was driven by the need to improve the sample environment ahead of a beamline optics upgrade which will see the size of the focal spot at the sample reduced to ~3×5 microns.
The new end station encompasses almost all aspects of the beamline that users interact with, and facilitates different modes of data collection. The horizontal goniometer is dedicated to in situ crystallography, allowing data collection from crystals held in crystallisation plates. In situ crystallography can provide extremely valuable feedback in the early stages of a project, removing conflating factors such as cryo-cooling and crystal handling when assessing crystal quality. It is also possible to collect wedges of data from multiple in situ crystals and merge these to obtain a complete dataset, an approach that has been extremely successful in, for example, the field of virus crystallography at I24.

Figure 1: Principal Beamline Scientist Robin Owen on the new I24 end station showing the two goniometers. The horizontal goniometer (for in situ experiments) is in the ‘in’ position while the vertical goniometer is retracted.
The vertical goniometer is dedicated to more conventional pin-based cryocrystallography. The orientation of the axis helps with stability and allows a small sphere of confusion (<< 1 micron) to be easily achieved. A kappa mount for reorientation of samples can be added if required with almost no impact on the sphere of confusion. Users can switch between modes in an automated fashion in a few minutes, allowing both types of experiment to be carried out in a single shift: a stark contrast to the previous end station. A concomitant upgrade of motor control hardware behind the scenes allows fast coordinated motions of multiple axes for high-speed grid and line scans in both modes.
Another aspect of the end station addressed during the upgrade is sample visualisation. The new on-axis viewing system offers greatly improved imaging of samples, with high resolution achieved at high zoom and a wide field of view at low zoom. A second viewing system allows very low zoom viewing of samples which often proves extremely useful for in situ experiments.
Taken together the changes to the end station will allow the reduced beamsizes now available at I24 to be fully exploited, and microfocus MX experiments to push new limits at Diamond.
Since September 2015, Diamond has a new facility for supporting compound development for medicine and biology: the XChem facility is a world-wide first for synchrotrons, providing access to crystal-based fragment screening. Attached to MX beamline I04-1, the facility provides access to a highly streamlined process, allowing users to test an unprecedented 1,000 compounds individually in a single week – order of magnitudes faster than possible by old technology.
The screening experiment’s aim is to identify hits that can be developed to lead drug candidates or other molecules that exhibit biological action. Fragment-based methods in drug discovery are a well-established approach, which emerged as an alternative to traditional and infrastructure-heavy high throughput screening. “Fragments” are the name for small compounds that tend to bind in a drug-like way to many biological targets, so this approach guarantees finding hits. The tradeoff is that affinity between fragments and their target proteins are generally weak, so highly sensitive analytical techniques are needed to detect their binding. X-ray crystallography is the most sensitive of all, but had been largely abandoned as a screening technique as it was too expensive and time consuming; Diamond’s XChem facility has now changed the game.
The process provided by XChem covers several aspects:
The only important bottleneck that remains is crystal preparation, which is the users’ responsibility; but the support team (I04-1 and Industry Liaison) are able to provide expert advice.
General access to XChem was successfully trialed from late 2014, and friendly early users were crucial to establishing how to roll out the current fully fledged user program; the facility was officially launched in November 2015. To date, over a dozen external experiments have been completed by users from both academia and industry (Fig. 3). The earliest external user, Chris Schofield’s group at Oxford University, has already been able to use the results for successfully developing inhibitors against proteins of antibiotic resistance.
 |  |
| Figure 2: Left: I04-1 PDRA Patrick Collins working in the XChem lab. Right: Beamline I04-1 experimental hutch. | |
The XChem facility is the outcome of a long-term partnership with Oxford University; I04-1’s Principal Beamline Scientist, Frank von Delft, is also jointly head of the Protein Crystallography group at Oxford’s SGC, with the SGC groups of Paul Brennan and Brian Marsden collaborating closely. Additionally, Oxford's EPSRC and MRC Systems Approaches to Biological Science (SABS) Centre for Doctoral Training has contributed several outstanding students, whose highly original contributions have been crucial to the viability of the facility.
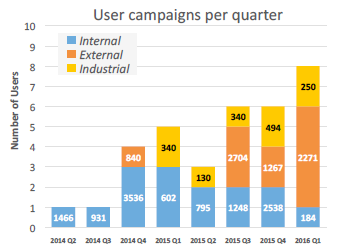
Figure 3: Screening experiments performed to date at the XChem facility; numbers on the bars indicate how many crystals have been screened.
The last decade has witnessed remarkable advances in photon counting detectors, micro-, multi-crystal and serial crystallography and serial femtosecond crystallography using X-ray free electron lasers (XFELs) (the latter accessed through Diamond’s XFEL Hub). The pace of change and new types of data emerging has created a need and opportunity for new state-of-the-art analysis software.
DIALS was conceived to address these challenges and is a collaboration between Diamond Light Source, CCP4 (the BBSRC-funded Collaborative Computational Project No. 4), Lawrence Berkeley National Laboratory and the MRC Laboratory of Molecular Biology in Cambridge. It has been funded from the EU project BioStruct-X.
DIALS is a modular, extensible, software toolkit for both developers and users and is enabling Diamond to respond quickly to new developments in the field, for example the measurement of weak, serial crystallography data from nanocrystals. It enables the rapid prototyping and development of new algorithms for data analysis and is encouraging strong collaboration amongst a number of UK, European, US and Japanese groups due to its being completely open source and free to use.
Amongst its novel features are the ability to refine and analyse data from multiple crystals simultaneously, benefitting from commonality in experimental parameters, and the development of algorithms specifically for weak, low background data from photon counting detectors. The software is already benefitting the structural biology community by producing higher resolution data and structures both from synchrotrons and XFELs.
Early in 2016 DIALS was officially released to the community within CCP4 and Phenix crystallography packages. In many respects DIALS is still in its infancy and the team are excited about the many new opportunities that it opens up.
The user community comes from industry and academia and makes up around 60% of all Diamond users. Studies are focused on structural biology, ranging from drug discovery to fundamental biology. An overall trend to faster, more efficient operation means that experiments can now be completed in two minutes; this has transformed what is possible. In 2015, experiments performed on the village’s five operational beamlines led to 378 peer reviewed papers and 651 protein data bank entries.
Since operations began, all beamlines have moved to greater automation and now more than 50% of user visits are performed remotely. Automation of sample presentation has been a priority over the last year. The new robotic sample changer, BART, became operational in January 2015 on beamline I03, a development led by the I03 beamline team. This version initially could hold 368 samples. A new version of the sample changers, BART 2, is capable of holding 592 samples and these were installed on beamlines I04 in September and I04- 1 in November 2015. An upgrade in January 2016 to BART on I03 raised its capacity to 480 samples. These new systems now give sample exchange times between 17 - 22 seconds. A Bart 2 robot system will be installed on beamline I24 in 2016. This automation has greatly increased sample throughput and allows better exploitation of valuable beamtime.
2015 saw the preparation of a new versatile MX in situ beamline (VMXi), that will replace beamline I02 in 2016. The new beamline will allow data to be collected from crystals at room temperature in their crystallisation media, rather than under cryogenic conditions (as with I02) allowing for the study of crystals at their earliest point and the collection of data from all crystals, including those that cannot be studied at very low temperatures. The new facility available to users in 2016 will have automated transfer between sample storage and the beamline, using the BART robot control system, as well as automated data collection and screening. It will in effect be userless, delivering rapid results to users without the need for their participation directly in the X-ray experiment.
Beamlines I03 and I04 provide a similar service to I02 but in addition, I03 is the only beamline in Europe (and the only automated beamline worldwide) that can provide containment measures for experiments involving biological agents in hazard group 3. This includes dangerous bacteria, parasites and viruses, where the knowledge of the 3D structures of biological molecules can have a profound impact. I04 uniquely operates with compound refractive lenses focusing optics providing a range of beam sizes from the microfocus (5 micron) to larger beams (100 microns) over a wide energy range. When coupled with the use of multi-axis sample rotation and intelligent data collection, it can be used to solve difficult structures such as the viral protein structures solved by the Harvard group of Dr Donald Coen. Their experiments on beamline I04 enabled the solution of the structure of the human cytomegalovirus nuclear egress complex, a complex composed of two proteins connected by a viselike mechanism which helps the virus enter a cell’s nucleus1. Such structural information provided by Diamond could be important for the rational design of antiviral drugs.
The village also includes beamline I04-1, a fixed wavelength monochromatic beamline. It is ideal for routine data collection and high-throughput screening from well-diffracting crystals. As of April 2015, the beamline offers the world’s first facility for fragment screening by crystal structure. This drug discovery technique allows probing a potential drug target for drug binding sites by directly visualising how large numbers of very small molecules bind to the protein. The technique is described in more detail on the technical development tab on this page.
The microfocus beamline, I24, makes use of a two-stage demagnification design to provide a tuneable microfocus high flux beamline. The flexible beam properties and high flux densities available provide the ability to tackle microcrystals and other challenging samples down to a micron in size. Its automation is advanced, enabling sample loading, data collection and analysis. Recent upgrades to the beamline are discussed further on the technical development tab. The impact of the micro beam is illustrated by its ability to solve crystal structures from inside live cells as seen in a 2015 study by the group of Dr Robert Robinson from the Agency for Science, Technology and Research in Singapore. They used crystals 50 μm in length, formed inside a mammalian cell, to determine the crystal structure of PAK4, a kinase enzyme whose overexpression has been associated with some cancers. The authors described its binding to a potent kinase inhibitor, a protein class with growing potential for cancer therapeutics2.
Launched in February 2016, the I23 long wavelength beamline is a unique MX beamline optimised for the long wavelength region (1.5 – 4 Å). Compared to shorter wavelength diffraction, longer wavelengths will increase the contribution of anomalous diffraction arising from sulphur and phosphorus in native protein and DNA crystals to directly solve the crystallographic phase problem without additional labelling. Operation at long wavelengths presents some unique challenges to the sample environment, cooling and detection systems. The detector is semi cylindrical around the sample to accommodate large diffraction angles. The in vacuum sample environment eliminates air absorption, but requires conductive sample cooling during the sample transfer and experiments through specially designed sample mounts and cooling links to a pulse tube cryocooler3. This facility will be uniquely placed for researchers from the UK and worldwide community to solve structures from the most challenging targets which are not amenable to the facilities elsewhere at Diamond or at other synchrotrons.
The MX Village is now midway through construction of its new submicron beamline (VMXm) with optics installation due to start mid 2016. The beamline will enable routine structure determination from micro and nanocrystals that give rise to very weak diffraction. This is typically the case for crystals of flexible, complex biological macromolecules. The minimum X-ray beam size on VMXm will be less than 0.5 microns and will uniquely combine scanning electron microscopy with X-ray diffraction, to allow nanocrystals to be sequentially aligned into the X-ray beam for data collection. The beamline will be operational in January 2018.
The MX Village also includes the Full Field Cryo-X-ray Microscopy (B24) beamline. B24 is designed specifically to meet the rising demand for tomographic imaging of biological specimens under near physiological conditions and to bridge the resolution gap between electron microscopy and conventional light microscopy. The beamline is used for collecting data on cells, but also bacteria and lipid structures. At present B24 is operating in the water window – an X-ray energy range where water/ice is relatively transparent unlike carbon and other elements found in biological specimens which are strongly absorbed. As a result natural contrast occurs allowing imaging as close to the native state as it is possible to be. It is planned to extend the beamline’s operation to higher X-ray energies to allow data to be collected on thicker specimens.
References:
Diamond Light Source is the UK's national synchrotron science facility, located at the Harwell Science and Innovation Campus in Oxfordshire.
Copyright © 2022 Diamond Light Source
Diamond Light Source Ltd
Diamond House
Harwell Science & Innovation Campus
Didcot
Oxfordshire
OX11 0DE
Diamond Light Source® and the Diamond logo are registered trademarks of Diamond Light Source Ltd
Registered in England and Wales at Diamond House, Harwell Science and Innovation Campus, Didcot, Oxfordshire, OX11 0DE, United Kingdom. Company number: 4375679. VAT number: 287 461 957. Economic Operators Registration and Identification (EORI) number: GB287461957003.