The trans-effect is a well-known phenomenon in coordination chemistry relating to organic ligand substitution. When an organic ligand is bound to a metal centre, it can cause the ligand directly opposite it (in the trans position) to be more liable to substitution, which in turn can change the charge of the compound. It applies to organic ligands, which tend to exhibit strong electronic properties. Affecting both square planar and octahedral complexes, the trans-effect can be exploited to create compounds of a particular desired structure.
Recently researchers have suggested that metal surfaces coordinated to a metal-organic complex could behave in a similar way. In a study published in Chemical Science, an international group of researchers have tested this theory and provided evidence showing that metal surfaces can indeed exhibit trans-effects. The team used the X-ray Standing Waves (XSW) technique at the Surface and Interface Structural Analysis beamline (I09) to accurately measure bond distances when a metal-organic complex was adsorbed onto a metal surface. Their results corroborated the idea that the metal-surface bond length changes depending on which ligand is bound at the trans position, proving the so-called ‘surface trans-effect’.
The researchers supported their observations with theoretical Density Functional Theory (DFT) calculations, which showed that the metal surface was able to act as a trans-directing ligand. Their studies also showed that the surface trans-effect is delocalised throughout the metal surface, rather than being a localised atomic phenomenon.
The trans-effect
The trans-effect is caused by organic ligands with strong σ-donor or-π acceptor electronic properties. Strongly trans-directing ligands weaken the ligand-metal bond that lies directly opposite, in the trans position. This causes the metal-ligand bond length in the trans position to lengthen, and thus become more liable to substitution.
Recently researchers have postulated that metal surfaces, when bound to metal-organic complexes, could also act like ligands, and demonstrate a similar trans-effect. Termed the surface trans-effect, evidence has been found that adsorbate supporting metal surfaces can affect the electronic properties of an adsorbed metal coordinated centre. Researchers predict that, should the surface trans-effect exist, it would cause the bond length between the coordinated metal centre and surface it is adsorbed to change depending on the ligand in the trans position but until now this had not been measured.
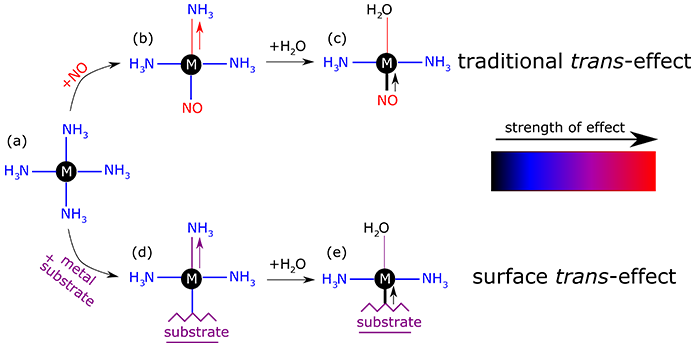
Figure 1: Illustration of the traditional trans-effect compared to the surface trans-effect for a hypothetical system. Just as replacing (a-b) a weak trans ligand, like ammonia (NH3), with a strong trans ligand, like nitric oxide (NO), results in a longer bond opposite the NO; replacing (a-d) a weak trans ligand (NH3) with a substrate that has a stronger trans-effect results in a longer bond opposite the surface. In turn, if you replace (b-c and d-e) a weak trans ligand (NH3) with a ligand with an even weaker trans-effect, like water (H2O), then you will shorten the bond that is opposite the H2O.
Measuring bond distances
To resolve the bond distances, an international team of researchers from Diamond and universities in Germany, France, the UK, and the USA, turned to the X-ray Standing Wave (XSW) technique at Diamond’s I09 beamline.
The XSW technique allowed the team to accurately measure the bond distances between a metal surface and variously substituted metal-organic complexes, even though the concentration of complex bound to the surface was very low. It also provided a way of measuring distances in a system that was vertically but not laterally ordered, as while the metal centres were a similar distance from the surface they did not adsorb into a well-defined site on the surface.
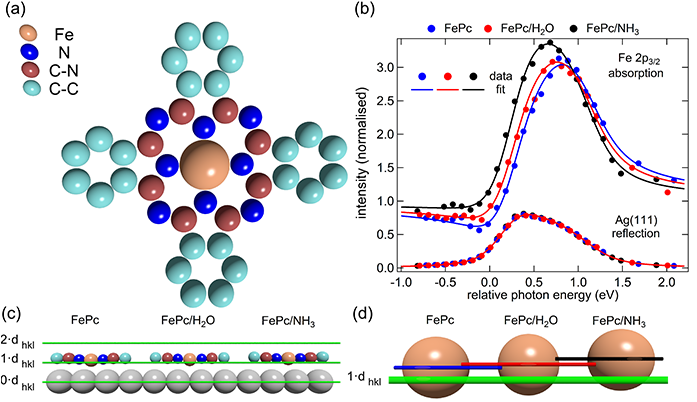
Comparing to theory
The team compared their measurements to those they calculated theoretically by DFT and found a remarkable similarity between the two.
The DFT calculations allowed the team to map the electron density around the metal centre - first for FePc adsorbed to the Ag surface alone, and then with ammonia and then water bound in the trans position. They also carried out calculations on FePc coordinated to various ligands, when not adsorbed onto the Ag surface.
“We looked at FePc adsorbed to Ag and saw electrons pushed along the sigma direction. The shape of the bonding was very similar to the other complexes, so that told us that it’s actually a trans-effect,” said David Duncan, the beamline scientist at I09 who was part of the research team.
A surface phenomenon
The team also found that the trans-effect was not coming from a single surface atom but from the whole surface: “Experimentally the molecule we put on the surface has no registry with the atoms below it, which means that it doesn’t have a well defined location. Some of the molecules might have a silver atom below them, some might be bridging two silver atoms, some might have an arbitrary position,” said Duncan.
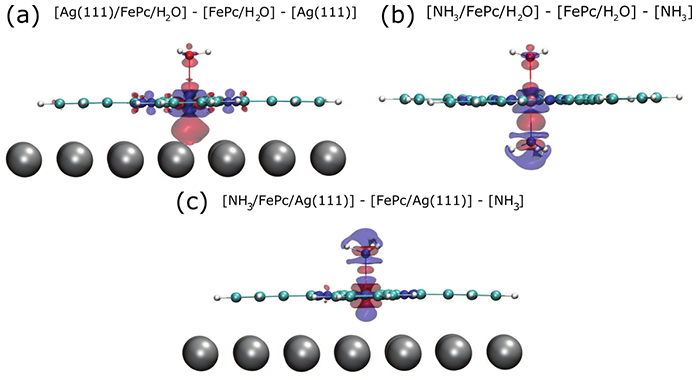
Figure 3: The redistribution of the electrons around the iron centre of the molecule due to introducing the (a) silver surface or (b) an ammonia molecule opposite to a water molecule. The introduction of both the silver surface and the ammonia resulted in a very similar change along the water to iron to silver/ammonia bonding direction – indicative of a trans-effect. Also shown (c) is the effect of introducing the silver surface opposite to ammonia, resulting in a very similar “parachute” shape around the hydrogen atoms of the ammonia molecule.
Future work
Although the team’s work is inherently fundamental, they hope this additional understanding of the surface trans-effect can be used in other areas of research. Metal phthalocyanines can be used as gas sensors, as they display distinct electronic changes upon ligation to different gases. The surface trans-effect suggests that this ligation would have a direct effect on the metal substrate, enchancing the signal of the gas sensor. However, for metal-organic catalysts adsorbed to a surface the surface trans-effect would suggests a drastic change in activity due to the underlying substrate, which may require adsorbed metal-organic catalysts to be made of more reactive materials.
To find out more about the I09 beamline, or to discuss potential applications, please contact Principal Beamline Scientist Dr Tien-Lin Lee: [email protected].
Diamond Light Source is the UK's national synchrotron science facility, located at the Harwell Science and Innovation Campus in Oxfordshire.
Copyright © 2022 Diamond Light Source
Diamond Light Source Ltd
Diamond House
Harwell Science & Innovation Campus
Didcot
Oxfordshire
OX11 0DE
Diamond Light Source® and the Diamond logo are registered trademarks of Diamond Light Source Ltd
Registered in England and Wales at Diamond House, Harwell Science and Innovation Campus, Didcot, Oxfordshire, OX11 0DE, United Kingdom. Company number: 4375679. VAT number: 287 461 957. Economic Operators Registration and Identification (EORI) number: GB287461957003.