Fungus-like eukaryotic plant pathogens of the genus Phytophthora are devastating to root crops and vegetables because the effector proteins they produce can evolve rapidly to evade recognition by the host’s immune system.
Until now the molecular mechanisms underlying this evolutionary arms race have been poorly understood. New research by a team from the BBSRC’s John Innes Centre, Sainsbury Laboratory and the University of East Anglia, have used Diamond’s MX beamlines to shed light on the structures and processes involved.
The team studied crystal structures of the effector domains from two such proteins, one from Phytophthora infestans, the potato blight pathogen infamous for causing famine in Ireland, and one from its relative Phytophthora capsici, which attacks cucumbers and squashes.
Their findings, published this month in the Journal of Biological Chemistry shows whilst proteins from related species differ widely in their effector domains, they share a conserved core α-helical fold. As shown in the figure below, this provides structural integrity within and around which the protein shows the evolutionary plasticity that enables it to disguise itself from host immune systems.
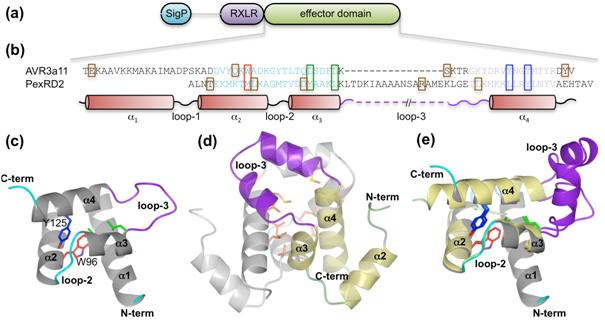
The core α-helical fold occurs in 44% of Phytophthora RXLR effectors, though these show less than 20% sequence identity in their effector domains. This suggests that the core fold is an ancestral structural ‘scaffold’ that enables functional adaptation of these fast-evolving effectors through insertion/deletions in loop regions between α-helices, extensions to N- and C-termini, amino acid replacements in surface residues, tandem domain duplications and oligomerization.
This may have important implications for crop protection and the potential development of effective treatments.
Structures of Phytophthora RXLR effector proteins: a conserved but adaptable fold underpins functional diversity, Laurence S. Boutemy, Stuart R. F. King, Joe Win, Richard K. Hughes, Thomas A. Clarke, Tharin M. A. Blumenschein, Sophien Kamoun and Mark J. Banfield, Journal of Biological Chemistry (2011) DOI: 10.1074/jbc.M111.262303
Diamond Light Source is the UK's national synchrotron science facility, located at the Harwell Science and Innovation Campus in Oxfordshire.
Copyright © 2022 Diamond Light Source
Diamond Light Source Ltd
Diamond House
Harwell Science & Innovation Campus
Didcot
Oxfordshire
OX11 0DE
Diamond Light Source® and the Diamond logo are registered trademarks of Diamond Light Source Ltd
Registered in England and Wales at Diamond House, Harwell Science and Innovation Campus, Didcot, Oxfordshire, OX11 0DE, United Kingdom. Company number: 4375679. VAT number: 287 461 957. Economic Operators Registration and Identification (EORI) number: GB287461957003.