Dr Edwin Yates, University of Liverpool
The two prototypical members of the Fibroblast growth factors (FGF) family, FGF-1 and-2, have fundamentally distinct binding requirements and the secondary structures induced by binding of polysaccharides are also distinct. Analogues of HS, which are active with FGF-1 act in essentially the same way as the natural ligand, but not in the case of FGF-2. This improves our understanding of the structural basis of the signalling complexes and is vital for the design of analogues, which can be developed to influence signalling processes selectively in disease states. Complementary spectroscopic techniques in the Diamond support laboratory (near UV-CD, Raman and Raman optical activity) were also recorded for a series of related compounds, HS, heparin and analogues. These were the first recorded spectra in many cases, providing initial assignments, for these molecules, and these results lay the foundation for further in-depth studies, which will provide detailed conformational information for the first time.
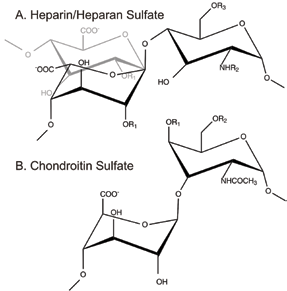
![Figure 1: Schematic Illustration of the glycosaminoglycans (A) heparan sulfate [-4) α-L-IdoA (grey)/β-D-GlcA (black) 1-4 α-D-GlcN (1-] and its analogue heparin [-4) α-L-IdoA 1-4 α-D-GlcN (1-]( where R1 = H or SO3 -, R2 = H/ COCH3 or SO3 - and R3 = H or SO3 -). and (B) chondroitin sulfate [→4) D-GlcA β (1→3) D-GalNAc β (1→], where R1 or R2 = H or SO3 -.](/dam/jcr:fba26c40-c676-4321-a9a2-0f7851417348/Yates1.2020-02-21-14-24-33.bmp) |
| Figure 1: Schematic Illustration of the glycosaminoglycans (A) heparan sulfate [-4) α-L-IdoA (grey)/β-D-GlcA (black) 1-4 α-D-GlcN (1-] and its analogue heparin [-4) α-L-IdoA 1-4 α-D-GlcN (1-]( where R1 = H or SO3 -, R2 = H/ COCH3 or SO3 - and R3 = H or SO3 -). and (B) chondroitin sulfate [→4) D-GlcA β (1→3) D-GalNAc β (1→], where R1 or R2 = H or SO3 -. |
FGFs are a family of key regulatory molecules present on the surfaces of all mammalian cells and are involved in normal, healthy development, cell growth and wound healing. More sinister, however, is their involvement in cancer-related processes, particularly the development of new blood vessels (angiogenesis) in tumours. Influencing FGF signalling selectively is a long-held goal with considerable medical promise. In earlier work, we showed that modified heparin polysaccharide analogues based on heparin. were capable of differentially influencing FGF signalling and through FGF-2, also affecting angiogenesis [1]. Further progress has been hampered by a lack of detailed knowledge concerning the structural characteristics and interactions of FGFs, as well as the difficulty of developing and testing the effects of agents with the potential to moderate their activities. FGFs signal via formation of a complex containing one of four FGF receptors but, only do so in the presence of a suitable, naturally occurring polysaccharide, which is provided in vivo by heparan sulfate (HS) [2].
As part of our on-going researches in this area, we have developed a series of analogue polysaccharides with the ability to support FGF signalling selectively, while lacking many of the undesirable activities associated with the naturally occurring polysaccharide, such as bleeding. For many proteins, the obvious way to gain detailed structural information concerning their interactions would be to study the protein structure using X-ray crystallography, for which Diamond would be ideally suited but, the need to study interactions between the protein and these polysaccharides which do not crystallise precludes this possibility. However, the versatility of the facilities at Diamond have proved equal to the task. In collaboration with Prof. Giuliano Siligardi and using Diamond’s Circular Dichroism beamline B23, we have begun to explore some of the detailed structural interactions of the two principal FGF proteins, FGF-1 and FGF- 2, as well as their interactions with both natural polysaccharide ligands and synthetic analogues (Fig. 2).
 |
| Figure 2: SRCD spectra (180 - 260 nm) of FGF-1 (A) (recorded at 0.9 mg/ml in phosphate buffer) and FGF-2 (B) (recorded at 0.5 mg/ml in phosphate buffer) with a selection of sulfated plant polysaccharides (at 1/5th molar equivalence of the polysaccharide with respect to the protein). All spectra were normalised at 260 nm and smoothed using the Savitzky-Golay method, 12 point window and second order polynominal. |
Circular Dichroism (CD) arises from the difference in absorbance by a chiral structure of right and left circularly polarised radiation. In the ultraviolet region (180-250 nm) CD is able to distinguish protein secondary structural features, for example, ‘ helices, ‘ sheet, turns and un-ordered stretches. This is achieved essentially by its ability to measure differences in absorbance between electronic transitions (n to π* and π to π*) in the amide bonds of the peptide backbone, which has subtly different spatial arrangements in each of the secondary structural types and, when measured over the range 180-250 nm, exhibits characteristic curves. Moreover, CD provides a sensitive measure of changes in these secondary structural features for proteins and, importantly, can do so for proteins interacting with polysaccharides in solution: an environment more closely resembling that found at the cell surface. CD provides a convenient means by which inhibitors selective for one protein, but not another, may be identified. For both the natural polysaccharide ligand and any potential analogues, it is important to know what the effect of binding is on the structure of the protein.
The two prototypical members of the FGF family, FGF-1 and -2, have fundamentally different binding requirements and the secondary structures induced by the binding of polysaccharides are also distinct. Some polysaccharide analogues, which were active with FGF-1, were shown to influence the secondary structures of FGF-1 in essentially the same way as the natural ligand [e.g. P10 in Figure 2A], but this was not the case with FGF-2, with which interactions caused distinct structural changes [Figure 2B]. These results from B23 help improve our understanding of the structural basis of FGF signalling complexes and provide an additional means by which the structural impact of potential inhibitors can be assessed. This will be vital to the future design and screening of improved analogues with the ultimate aim of being able to influence signalling processes selectively in disease states [3].
![Figure 3: Raman spectra [500-2000 cm-1] (IR+IL) of: (a) (upper) heparin (porcine mucosal, 1 g.ml-1), (b) (upper) N-acetylated heparin (0.5 g.ml-1), (c) (upper) HS (porcine 0.2 g.ml-1), (d) (upper) CS (0.5 g.ml-1). Back scattered Raman optical activity spectra (IR-IL) of: (a) (lower) heparin (porcine mucosal, 1 g.ml-1), (b) (lower) N-acetylated heparin (0.5 g/ml), (c) (lower) HS (0.2 g.ml- 1), (d) (lower) CS (0.5 g.ml-1).](/dam/jcr:fba26c40-c676-4321-a9a2-0f7851417348/Yates1.2020-02-21-14-24-33.bmp) |
| Figure 3: Raman spectra [500-2000 cm-1] (IR+IL) of: (a) (upper) heparin (porcine mucosal, 1 g.ml-1), (b) (upper) N-acetylated heparin (0.5 g.ml-1), (c) (upper) HS (porcine 0.2 g.ml-1), (d) (upper) CS (0.5 g.ml-1). Back scattered Raman optical activity spectra (IR-IL) of: (a) (lower) heparin (porcine mucosal, 1 g.ml-1), (b) (lower) N-acetylated heparin (0.5 g/ml), (c) (lower) HS (0.2 g.ml- 1), (d) (lower) CS (0.5 g.ml-1). |
The effectiveness of the experimental work on B23 was considerably enhanced by the availability of complementary spectroscopic techniques in the laboratory (particularly near UV-CD, Raman and Raman optical activity [ROA]). These techniques promise to shed new light on the structural features of polysaccharides, and include one of the few ways of directly observing helicity in polysaccharides [4]. The Raman optical activity (ROA) spectra shown in Fig. 3 were the first ROA spectra recorded for these molecules and provide initial assignments [5]. These results lay the foundation for further in-depth studies, which will provide detailed conformational information concerning the polysaccharides.
Future research will concentrate on combining spectroscopic techniques, for which the facilities at Diamond are well-designed, shedding further light on both the protein and polysaccharide components of this challenging system and bringing the goal of selective inhibitors of FGF signalling, with the promise of considerable medical benefits, nearer.
References
[1] S.E. Guimond, J.E. Turnbull, E.A. Yates, Engineered bioactive polysaccharides from heparin. Macromol Biosci, 6 (8), 681-6, (2006).
[2] A. Ori, M.C. Wilkinson, D.G. Fernig, The heparanome and regulation of cell function: structures, functions and challenges. Front Biosci, 13, 4309-38 (2008).
[3] T.R. Rudd et al., Degeneracy in the structure and activities of heparin derivatives paves the way for active non-GAG analogues with FGF signallings, Macromol Biosci, (2010).
[4] A.F. Bell, L. Hecht, L.D. Barron, Polysaccharide vibrational Raman optical activity: Laminarin and pullulan. J Raman Spectrosc, 26 (12), 1071-1074 (1995).
[5] T. R. Rudd et al., Raman and Raman Optical Activity of Glycosaminoglycans, Chem. Commun., 46, 4124 - 4126, (2010). DOI: 10.1039/c001535k
Authors
T.R. Rudd, M. A. Skidmore, S. E. Guimond, R. Xu, R.Hussain, D. G. Fernig, G. Siligardi and E. A. Yates.
Funding Acknowledgement
Biotechnology and Biological Sciences Research Council, Medical Research Council and The Royal Society.
Diamond Light Source is the UK's national synchrotron science facility, located at the Harwell Science and Innovation Campus in Oxfordshire.
Copyright © 2022 Diamond Light Source
Diamond Light Source Ltd
Diamond House
Harwell Science & Innovation Campus
Didcot
Oxfordshire
OX11 0DE
Diamond Light Source® and the Diamond logo are registered trademarks of Diamond Light Source Ltd
Registered in England and Wales at Diamond House, Harwell Science and Innovation Campus, Didcot, Oxfordshire, OX11 0DE, United Kingdom. Company number: 4375679. VAT number: 287 461 957. Economic Operators Registration and Identification (EORI) number: GB287461957003.