Prof Stephen Curry, Imperial College London
The regulation of the alternative splicing of messenger RNA allows mammals and other eukaryotic organisms to greatly enrich the diversity of the proteins coded by their genomes. Most eukaryotic genes are fragmented into introns that need to be joined up before the encoded protein is synthesised. But by combining the introns in different ways, the cell can make different proteins from the same genetic material.
The control mechanisms for alternative splicing are not well understood though they are known to involve interactions between sets of regulatory proteins. We have been investigating the interaction between two proteins, Raver1 and PTB (Polypyrimidine Tract Binding protein), which co-ordinate the splicing of a muscle protein, α-tropomyosin. The proteins bind to one another and to RNA. The interaction between the proteins appears to be due to the presence of four PTB-binding peptide motifs within Raver1—two of high affinity and two of low affinity—which bind to PTB RRM2 (RNA recognition motif 2).
To examine how these peptides bind to PTB we used genetic engineering to fuse each peptide, via a long peptide linker, to the N-terminus of PTB-RRM2. Two of the resulting chimeras crystallised and allowed us to determine the high-resolution structures of the peptide-RRM2 complexes. Comparison of the structures revealed a surprising degree of plasticity in the conformation of the bound peptides, which may well account for the differences in their binding affinities. These results are helping us to perform a detailed structural and functional dissection of the Raver1-PTB interaction.
The mosaic structure of genes in mammals and other eukaryotes means that the pre-messenger RNA (pre-mRNA) transcribed from the DNA has to be spliced to remove the exons and stitch together the introns so that the mRNA can be translated to yield a functional protein. In many instances the cell can make decisions about what introns to retain in the final message. Such alternative splicing is a powerful mechanism for generating functionally different proteins from the same gene.
In the case of α-tropomyosin, introns 2 and 3 are mutually exclusive and the decision about which one to include is somehow determined by the concerted action of two proteins, Raver1 and PTB (Polypyrimidine Tract Binding protein), which interact with each other and with control sequences in the premRNA in the vicinity of the regulated introns [1].
PTB is an RNA-binding protein with four RNA recognition motifs (RRMs); Raver1 contains three RRMs at its N-terminus but has a large C-terminal domain with four conserved peptide sequence motifs that an bind PTB (Fig. 1a). Only two of these motifs (m1 and m3) appear to bind PTB with relatively high affinity. In earlier work using NMR we showed that these peptide motifs bind specifically to a single site on the dorsal, helical surface of PTB-RRM2 [2]. However, the binding affinity for a synthetic peptide corresponding to the m3 motif not sufficient (KD ~100 µ M) to permit a highresolution structure determination by NMR.
To circumvent this problem by artificially enhancing the peptide binding affinity, we generated chimeras in which the PTB-binding peptides from Raver1 were fused to the N-terminus of PTB-RRM2 (Fig. 1b). Although these chimeric constructs were designed for use in NMR experiments, the resulting proteins crystallised in two cases (m3 and m4; Fig. 1c). Crystals of the Raver1(m4)-PTB chimera diffracted to 1.55 Å on beamline I04 at Diamond and yielded a very high resolution electron density map (Fig. 1d) after phasing by molecular replacement (using our solution structure of RRM2 as a search model [3]).
The crystal structures confirm that the peptide-binding site is on the helical face of PTB-RRM2 on the opposite side of the domain to the RNA-binding β-sheet (Fig. 1e). Both of the m3 and m4 peptides from Raver1 bind to this site with broadly similar conformations but the crystal structures unexpectedly reveal that there are nevertheless significant differences in the ways that they associate with RRM2 (Fig. 1f,g).
Each of the Raver1 peptides adopts a pinched conformation that allows the central pair of Leu residues to insert their side-chains into a shallow hydrophobic patch between the two helices of RRM2. However, on either side of this central feature there are striking differences in the peptide backbone conformations. In the high-affinity motif, m3, a hydrogen bond from a Ser side-chain to the downstream backbone appears to stabilise the pinched peptide conformation. Moreover, the sequence LGEP wraps around the side-chain of Tyr 247 in PTB-RRM2. For the low-affinity motif, m4, there is no pinching hydrogen bond and the sequence of amino acids that wraps around Tyr 247 is only three residues long (LGL).
These differences mean that the peptides make distinct sets of hydrogen bonds with PTB-RRM2. It is apparent that the m3 peptide makes more hydrogen bonds than m4 (Fig. 1f,g) and this finding immediately suggests a new hypothesis to explain the differences in the affinities of the two motifs for PTB. We will test this by making binding measurements using peptides that are mutated at the positions observed to affect the interaction with RRM2.
This study—which is an ongoing collaboration with Chris Smith (Cambridge) and Steve Matthews (Imperial)—is a great example of the investigative power of high-resolution protein crystallography. Not only did it reveal a completely unanticipated degree of plasticity in an important protein- protein interaction, but it also provided us with precisely the information needed to test the new ideas about binding specificity that have arisen from the structure.
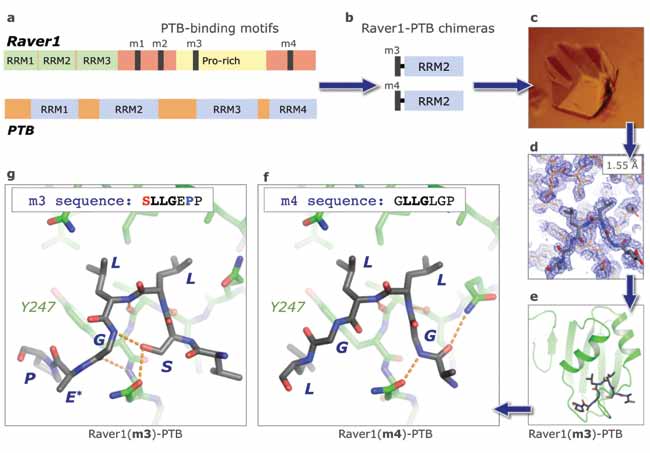 |
| Figure 1: The story of the structural investigation of the interaction between Raver1 and PTB. (a) Schematic representation of the domain structures of Raver1 and PTB. (b) Schematic representation of the Raver1-PTB chimeras that yielded diffraction-quality crystals. (c) Crystal of Raver1(m3)-PTB. (d) Electron density map calculated for Raver1(m4)-PTB using data collected on beamline I04 at the Diamond Light Source. (e) Overview of the structure of the Raver1-PTB ‘complex’. The linker connecting the Raver1 peptide to the N-terminus of PTB-RRM2 is disordered in the electron density map and therefore not included in the model. Close-up views of the peptide interactions with PTB-RRM2 for (f) Raver1 motif m3 and (g) Raver1 motif m4. (The residue labelled E* in panel g was actually mutated to Ala, but this makes no difference to the binding affinity). |
References
[1] Gromak, N., Rideau, A., Southby, J., Scadden, A. D., Gooding, C., Huttelmaier, S., Singer, R. H. & Smith, C. W. The PTB interacting protein raver1 regulates alpha-tropomyosin alternative splicing. EMBO J, 22, 6356-64 (2003).
[2] Rideau, A. P., Gooding, C., Simpson, P. J., Monie, T. P., Lorenz, M., Huttelmaier, S., Singer, R. H., Matthews, S., Curry, S. & Smith, C. W. A peptide motif in Raver1 mediates splicing repression by interaction with the PTB RRM2 domain. Nat. Struct. Mol. Biol. 13, 839-48 (2006).
[3] Simpson, P. J., Monie, T. P., Szendroi, A., Davydova, N., Tyzack, J. K., Conte, M. R., Read, C. M., Cary, P. D., Svergun, D. I., Konarev, P. V., Curry, S. & Matthews, S. Structure and RNA interactions of the N-terminal RRM domains of PTB. Structure (Camb,) 12, 1631-43. (2004).
Principal Publication and Authors
Amar Joshi, Olga Kotik-Kogan, Peter Simpson, Steve Matthews, Chris Smith, Stephen Curry
Funding Acknowledgement
Biotechnology and Biological Sciences Research Council, UK.
Research carried out at Diamond on I04 and at EMBL-DESY where the dataset for the Raver1(m3)-PTB chimera was collected on X13 (Dec 2008).
Diamond Light Source is the UK's national synchrotron science facility, located at the Harwell Science and Innovation Campus in Oxfordshire.
Copyright © 2022 Diamond Light Source
Diamond Light Source Ltd
Diamond House
Harwell Science & Innovation Campus
Didcot
Oxfordshire
OX11 0DE
Diamond Light Source® and the Diamond logo are registered trademarks of Diamond Light Source Ltd
Registered in England and Wales at Diamond House, Harwell Science and Innovation Campus, Didcot, Oxfordshire, OX11 0DE, United Kingdom. Company number: 4375679. VAT number: 287 461 957. Economic Operators Registration and Identification (EORI) number: GB287461957003.