The search for new and improved ion-exchange materials for nuclear waste remediation continues to be an important topic, especially given the issues over cleaning up existing legacy waste sitting in storage ponds, dealing with the reactors due to be decommissioned shortly and being prepared in advance to deal with radionuclides created by the new build of reactors recently announced. One family of materials that shows promise is layered metal phosphates, where cations such as Sr, Cs and Co can be trapped between the robust and stable layers. A detailed knowledge of the structures is crucial for a fundamental understanding of how these systems work and can be improved, so we have used Rietveld analysis of high resolution synchrotron X-ray powder diffraction data collected at beamline I11 to fully characterise the structural changes in systems of nominal composition Ti1‑xZrx(HPO4)2.H2O where x=0–1. The end members are isostructural systems that have known potential for the uptake in particular of Sr2+. We find that for x=0 to 0.3 and 0.9 to 1 the systems adopt the end member structures with subtle changes in the M–O distances; due to the very high precision of the results we can say that these are smaller than expected based on average metal ionic radii. The systems with x=0.4 to 0.8 exist as a mixture of Ti-rich and Zr-rich partially substituted systems. In combination with XRF results, we believe that there are two reasons why a complete solid solution does not form, firstly the structure appears to lack the flexibility to allow a smoothly changing average ionic radii without introducing strain that cannot, presumably, be relieved at the low synthesis temperatures and secondly that there is complex solution chemistry that prevents gel precursors with all ranges of compositions from forming. We have also used complementary pair distribution function analysis of high energy total X-ray scattering data collected at the APS to investigate whether or not there is any evidence of metal clustering, our results are only consistent with random distributions of the metal ions in the solids and not, as speculated in the literature, with crystallites having both regions of titanium phosphate and zirconium phosphate within the same particles.
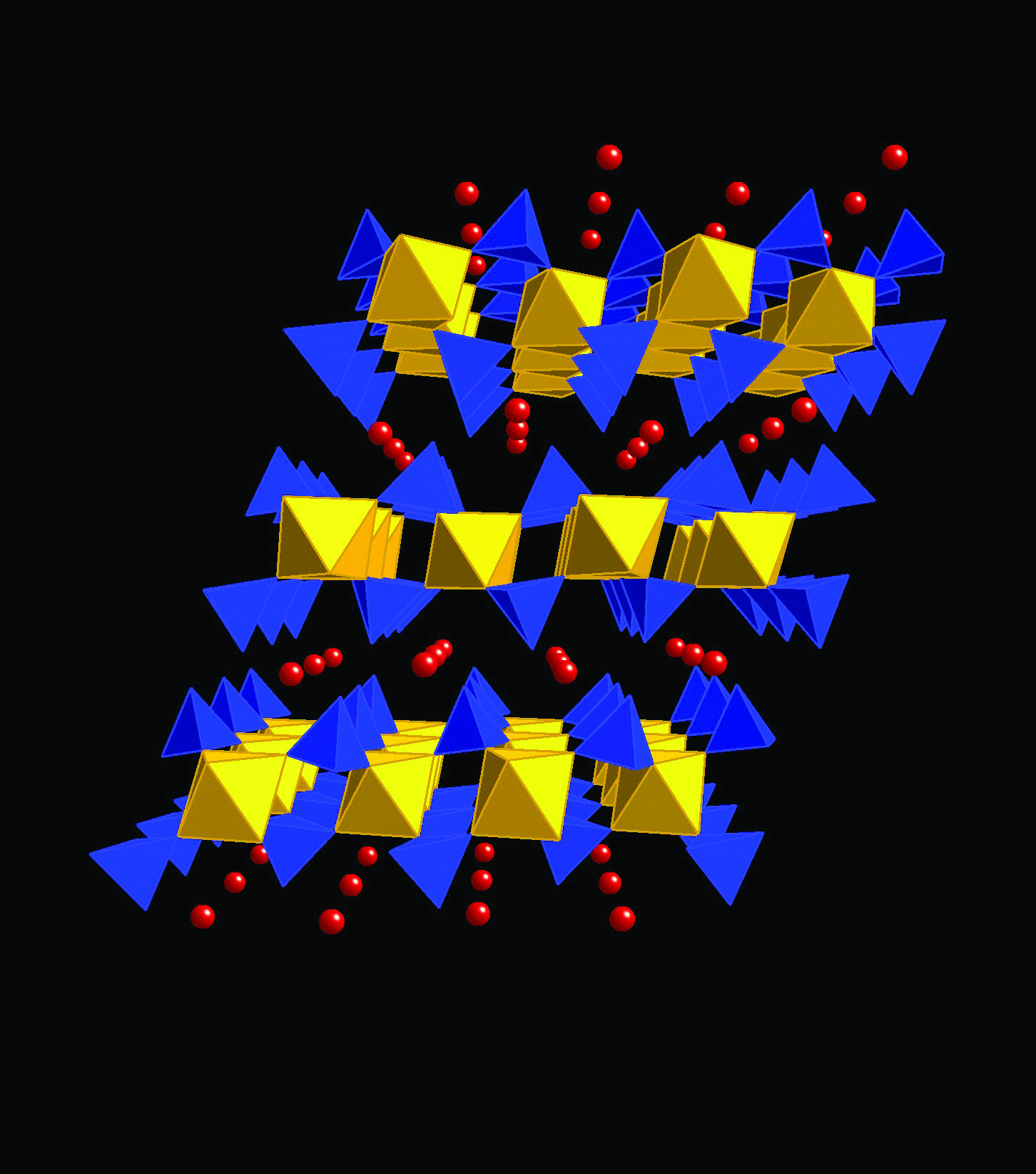
Figure 1: Polyhedral representation of α-Ti(HPO4)2×H2O with phosphate tetrahedra in blue, titanium octahedra in ochre and oxygen atoms of the intra-layer water molecules as red spheres. Hydrogen atoms have been omitted for clarity.
The ion exchange properties of the insoluble acid salts of tetravalent metal ions have been known for many years [1-6]. There has been a renewed recent interest in them owing to their favourable ion exchange properties and high resistance towards breakdown by temperature and/or radiation, making them ideal for use in the nuclear industry. The investigation of mixed metal compounds of this and other types is of interest because of the potential for fine tuning of the ion-exchange selectivity through chemical control. Our work focused on the so called alpha forms of the phosphates (α-MP), shown schematically in Fig. 1. In these systems, one oxygen atom of each of the phosphate groups that projects out between the layers is protonated. These acidic protons can be exchanged for other cations which will then locate between the layers and be bonded both to phosphate oxygen atoms and any intra-layer water molecules. The compounds that were the focus of this study have general formula Ti1-xZrx(HPO4)2.H2O, where x=0–1 and the end members are known to be isostructural. There are a few reported attempts to make the complete solid solution, but they have failed and no reason has yet been given for this. This is very unfortunate, because by changing the average radii of the metal species through doping Zr for Ti, one would make subtle changes in the structure that further alter the bonding environment between the layers where entering radionuclides reside, thus modifying the ion-exchange capabilities. In many cases, only small changes are necessary to turn a relatively poor ion-exchange system that leaches ions back into solution into a good one where very strong bonding occurs and the system is much more robust. Our goals include making a full analysis of detailed structural changes with doping that can then be correlated with aqueous ion-exchange studies to develop a comprehensive understanding. We also hope to produce a full solid solution or, failing that, use structural and chemical information to learn why compositions in the middle of the range will not form.
Materials spanning the composition range were made by a new hydrothermal route developed in our lab, but standard laboratory X-ray powder diffraction data was very complicated and could not be fully interpreted in many cases. To do this successfully, one needs to obtain powder diffraction data of the highest quality in terms of peak resolution and signal-to-noise, so we turned to the high resolution synchrotron X-ray powder diffractometer at beamline I11 and collected data sets as a function of composition on samples held in 0.5 mm diameter glass capillaries using the multi-analyser crystal detector array. In order to obtain complementary Pair Distribution Function data to probe any metal clustering, we also collected high energy total scattering data from beamline 11-ID-B at the APS.

Figure 2: Final observed (red crosses), calculated (green line) and difference (purple line) X-Ray diffraction profiles from the Rietveld refinement for α‑Ti(HPO4)2.H2O with reflection positions noted as vertical tick marks.
From the Rietveld analysis of the I11 data using GSAS [7], we were able to confirm that samples with x=0 to 0.3 and 0.9 to 1 were single phase with the known α-MP structure and obtained excellent information about the subtle changes in M–O bond lengths due to the high precision of the analysis. An example of the quality of fit is shown in Fig. 2 and a plot of the high precision average M–O distance for the single phase systems is shown in Figure 3. What is particularly interesting about the bond distances is that they do not obey a simple Vegard’s Law trend, i.e. they do not change in a linear fashion between the end members. In fact, it appears that they change little with metal doping in spite of the rather large differences in the ionic radii of Ti4+ and Zr4+, 74.5 and 86 pm, respectively. A consequence of this is that the structures will become increasingly strained and less stable as the composition changes from the end members towards the centre of the composition range. Given the superb high resolution of I11, we were also able to perform two-phase Rietveld refinements for the samples with x = 0.4 to 0.8 where there is so much peak overlap that analysis from even the best laboratory data will fail. These could all be analysed as mixtures of varying ratios of compounds of formula Ti0.68Zr0.32(HPO4)2.H2O and Ti0.14Zr0.86(HPO4)2.H2O. From the scale factors for the two phases we could gain useful estimates of the average compositions. In combination with XRF measurements, we were able to complete a picture that is fully consistent with a lack of flexibility within the octahedral metal coordination environment to changing size as expected with average ionic radius, and we believe this strain plays an important part in frustrating the formation of solids with the intermediate compositions. We also produced Pair Distribution Functions (PDF) for the complete range of materials from the high energy total scattering data collected at the APS. These were analysed using PDFGui [8] so that we could compare the refined structural parameters, the M–O bond lengths are also shown in Fig. 3 for comparison. In addition, through a consideration of the peaks due to M–M correlations, we could rule out previous suggestions that there were regions of Ti clustering and regions of Zr clustering; the data is fully consistent with a random solid solution.

Figure 3: Average metal-oxygen distance versus %Zr for the single-phase compositions in the Zr/Ti series. Red circles are data from the Rietveld refinements, blue squares from the PDF fits, the green line the expected values assuming Vegard’s Law and the red line the observed trend for the Ti-rich systems.
Our work has shed further light on the structural chemistry and solid solution limits of an important class of ion-exchange materials with potential utility for nuclear waste clean-up. We are now investigating how the ion-exchange for Sr2+ varies with composition and will correlate our new knowledge of the structural changes from the analysis of the I11 data with the observed chemistry. We are also tackling solving and refining the structures of the Sr-exchanged systems from a combination of high resolution synchrotron powder diffraction and PDF data, again collected at I11 and 11-ID-B, respectively.
References
[1] G. Alberti, P. Cardini-Galli, U. Costantino and E. Torracca, J. Inorg. Nucl. Chem. 29, 571-578 (1967).
[2] A. Clearfield, Inorganic Ion Exchange Materials, CRC Press, Boca Raton, FL, (1982).
[3] R. Llavona, M. Suarez, J. R. Garcia and J. Rodriguez, Inorg. Chem. 28, 2863-2868 (1989).
[4] A. Nilchi, M. Ghanadi Maragheh, A. Khanchi, et al., J. Radio. Nucl. Chem. 261, 393-400 (2004).
[5] A. Clearfield and T. N. Frianeza, J. Inorg. Nucl. Chem. 40, 1925-1932 (1978).
[6] A. Clearfield, W. L. Duax, A. S. Medina, et al., J. Phys. Chem. 73, 3424-3430 (1969).
[7] A. Larson, R. B. von Dreele, ‘GSAS: Generalised Structure Analysis System,’ Report No. L.A-UR-86-748: LANSCE, Los Alamos National Laboratory, Los Alamos, NM (1985).
[8] C. L. Farrow, P. Juhas, and J. W. Liu, J. Phys.: Cond. Matter. 19, 1-7 (2007).
Principal Publications and Authors
V.A. Burnell, J.E. Readman, C.C. Tang, J.E. Parker, S.P. Thompson, and J.A. Hriljac, Synthesis and Structural Characterisation using Rietveld and Pair Distribution Function Analysis of Layered Mixed Titanium-Zirconium Phosphates, Journal of Solid State Chemistry, (2010). DOI:10.1016/j.jssc.2010.07.028
Funding Acknowledgement
University of Birmingham
Research carried out on I11 at Diamond and the Advanced Photon Source.
Diamond Light Source is the UK's national synchrotron science facility, located at the Harwell Science and Innovation Campus in Oxfordshire.
Copyright © 2022 Diamond Light Source
Diamond Light Source Ltd
Diamond House
Harwell Science & Innovation Campus
Didcot
Oxfordshire
OX11 0DE
Diamond Light Source® and the Diamond logo are registered trademarks of Diamond Light Source Ltd
Registered in England and Wales at Diamond House, Harwell Science and Innovation Campus, Didcot, Oxfordshire, OX11 0DE, United Kingdom. Company number: 4375679. VAT number: 287 461 957. Economic Operators Registration and Identification (EORI) number: GB287461957003.