Dr Julia Richardson, University of Edinburgh
Transposons are pieces of DNA that can jump from one place to another in the genome of a cell and they make up a surprisingly large proportion of many genomes. In creating genetic diversity they have had a strong influence on the evolution of genomes. If they transpose they can directly affect gene expression, by inserting into genes or gene regulatory elements. However, their activity is more often suppressed by silencing mechanisms such as DNA methylation or RNAi. The mutational activity of transposons has facilitated the evolution of new genes and in some cases transposons have been domesticated by the host to provide new and beneficial cellular functions.
DNA transposons jump by a cut and paste mechanism mediated by a transposon-encoded transposase protein. This is achieved in an ordered series of events. We have determined the X-ray crystal structure of a transposition intermediate of the mariner transposon Mos1 (data collected at the ESRF and on Diamond Beamlines I04 and I03)The structure of the Mos1 paired-end complex (PEC), along with biochemical data, support a model for DNA binding where the transposase dimer holds the DNA ends in a parallel arrangement for cleavage. The architecture of this transpososome complex provides molecular insight into many facets of the Mos1 transposition mechanism: sequence-specific recognition of the transposon ends, the requirement for pairing of the ends prior to DNA cleavage, putative binding sites for flanking DNA and key molecular interactions that position the transposon ends for subsequent target DNA integration. A model for the target capture complex, supported by biochemical analysis of mutant transposases, reveals how the parallel arrangement of DNA in the Mos1 PEC facilitates integration of the transposon ends into positions separated by only 2 bp in target DNA. This structure increases our understanding of DNA transposition in general and of DNA rearrangements more broadly.
Genome sequencing efforts have revealed that transposable elements and their remnants make up a large proportion of many eukaryotic genomes (for example, ~45% of the human genome). The ability of transposons to integrate into genomes is being exploited to develop vectors for insertional mutagenesis, genome manipulation, transgenesis and gene therapy.
One class of transposable elements, DNA transposons, move from one genomic location to another by a cut and paste mechanism, mediated by transposon-encoded transposase proteins. Transposases are sequence-specific nucleases and DNA strand transferases that catalyze transposition through an ordered series of events: (a) sequence-specific binding of transposase to the terminal inverted repeats (IR) delimiting each end of the transposon, (b) pairing of the transposon IRs in a paired-end complex, (c) cleavage of one or both DNA strands at each transposon end, (d) capture of target DNA, and (e) strand transfer to insert the transposon at a new site.
DNA transposition is closely related mechanistically to the integration of HIV-1 and other retro-viruses as well as the DNA rearrangements involved in the generation of mammalian immune system diversity. The active sites that catalyse the strand cleavage and strand transfer reactions are highly conserved between DNA transposases and retroviral integrases and comprise a triad of acidic amino acids. These can bind two Mg2+ or Mn2+ ions [1], consistent with a twometal mechanism for DNA catalysis [2,3]. Structural analysis of DNA transposases has been limited and until recently, the only structures of intact transposase bound to IR DNA were for the bacterial transposon Tn5 [4].
Our goal is to investigate the molecular mechanism of the eukaryotic mariner/Tc1 family transposon Mos1. To this end we crystallised the paired-end complex formed from precleaved transposon ends and the Mos1 transposase. The crystals diffracted X-rays to a maximum resolution of 3.25 Å, and the structure was solved by molecular replacement, using our structure of the Mos1 transposase catalytic domain as the search model (PDB ID: 2F7T). The structure revealed two approximately parallel transposon ends bound to a dimeric enzyme (Figure 1, PDB ID: 3HOS and 3 HOT). The complex has a crossed (or trans) arrangement, with each transposon end recognised by the DNA binding region of one transposase monomer and by the active site of the other monomer. This crossed arrangement provides a molecular explanation for the requirement for pairing of the ends for DNA excision as it prevents undesirable chromosome breaks at a single end, before the transposon is committed to the transposition pathway.
All mariner/Tc1 family transposons integrate at TA target sites. In the Mos1 PEC structure the transposon 3’-OH ends are engaged in the active sites with the correct separation for integration into target phosphates separated by 2 base-pairs. Thus the crystallographic PEC appears to be in the correct configuration to carry out strand transfer. Based on the approximately parallel arrangement of the IRs, and analysis of transposase mutants in strand transfer assays, we presented a model for target DNA binding and a structural basis for integration into TA target DNA. Biochemical data provided support for this model and identified Arg186 to be critical for target integration.
Two additional DNA duplexes in the crystal indicated likely binding sites for flanking DNA. Each of these DNA duplexes links two separate PECs together in the crystal lattice, and is presumed to have been captured from the excess DNA in solution during crystallisation, thereby enabling crystal packing. The single orientation of each these extra duplexes with respect to the transposase active sites, was confirmed by the observation of one signal from iodinated DNA, with T16 in the IR DNA replaced by 5-Iodo-dU, in a Fo-Fc electron density map.
The Mos1 PEC structure has also provided insight into other members of the mariner/Tc1 family of transposons, including elements with promising biotechnology applications such as Sleeping Beauty.
| Figure A | Figure B |
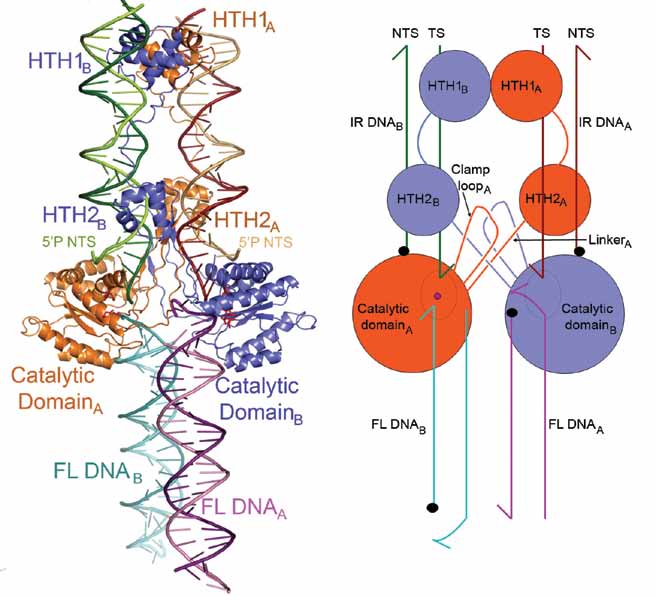 | |
| Figure 1. (A) The Mos1 PEC crystal structure: Transposase monomer A is coloured orange and monomer B blue. The two DNA duplexes bound by the N-terminal DNA-binding domains are labelled IR DNA and the two extra DNA duplexes are labelled FL DNA. (B) Schematic representation of the structure. The two helix-turnhelix (HTH) motifs in each DNA-binding domain bind in the major groove. The cleaved ends are bound by the catalytic domains. Arrows indicate 3’ ends and black dots the 5’ ends of DNA. | |
References
[1] Richardson,J.M., Dawson,A., O’Hagan,N., Taylor,P., Finnegan,D.J., & Walkinshaw, M.D. EMBO J., 25, 1324-1334. (2006).
[2] Beese,L.S. & Steitz,T.A. EMBO J. , 10, 25-33. (1991)
[3] Yang,W., Lee,J.Y., & Nowotny,M. Mol, Cell, 22, 5-13. (2006).
[4] Davies,D.R., Goryshin,I.Y., Reznikoff,W.S., & Rayment,I. Science, 289, 77-85. (2000).
Principal Publications and Authors
Richardson JM, Colloms SD, Finnegan DJ and Walkinshaw MD. Molecular architecture of the Mos1 paired-end complex:
the structural basis of DNA transposition in a eukaryote. Cell, 138(6), 1096-1108 (2009).
Funding Acknowledgement
The Wellcome Trust and the Medical Research Council, UK. Research carried out at Diamond on I03 and I04, and at the ESRF on BM14, ID23 and ID14-4.
Diamond Light Source is the UK's national synchrotron science facility, located at the Harwell Science and Innovation Campus in Oxfordshire.
Copyright © 2022 Diamond Light Source
Diamond Light Source Ltd
Diamond House
Harwell Science & Innovation Campus
Didcot
Oxfordshire
OX11 0DE
Diamond Light Source® and the Diamond logo are registered trademarks of Diamond Light Source Ltd
Registered in England and Wales at Diamond House, Harwell Science and Innovation Campus, Didcot, Oxfordshire, OX11 0DE, United Kingdom. Company number: 4375679. VAT number: 287 461 957. Economic Operators Registration and Identification (EORI) number: GB287461957003.