How can a disease that affects one species such as birds transfer to another such as humans? In the case of influenza, the answer could lie in a protein called hemagglutinin – the part of the flu virus that binds it to human cells when a person is infected. In the twentieth century there were three flu pandemics in 1918, 1957 and 1968. Scientists from the Medical Research Council have been using Diamond to study the structure of hemagglutinin from the virus strain that caused the 1957 “Asian” pandemic compared to the 1918 and 1968 outbreaks to try and discover why some avian flu viruses are more able to jump the species gap than others. This may help identify which avian flu viruses are more likely to bind to human cells and may help in preparations for future pandemics. Their research has been published in the journal PNAS.
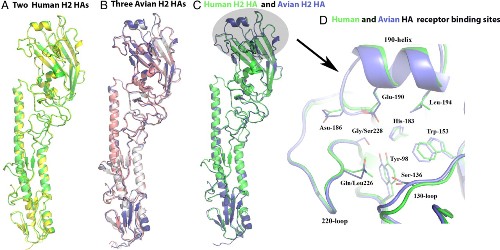 |
| Ribbons representation of different H2 HA monomers and receptor binding sites. |
The Asian flu pandemic of 1957, the second great flu pandemic of the 20th century, was caused by viruses that were a reassortment of human and avian, belonging to the H2N2 subtype. This virus was antigenically different from the H1N1 viruses of the Spanish influenza outbreak in 1918. Hemagglutinin is a receptor binding glycoprotein which binds to sialic acid. In birds, hemagglutinin prefers the sialic acid present in the intestine, but in humans it prefers a receptor that is predominantly in the upper respiratory tract. This implies that there has been a change in the receptor binding specificity.
The researchers used X-ray crystallography to examine the structural significance of this change by studying human and avian H2 virus in complex with analogues of the human and avian receptors. The study showed considerable similarity between the human and avian H2 hemagglutinins. Comparisons with the hemagglutinin H1 from the 1918 flu and the H3 hemagglutinin from the 1968 Hong Kong strain showed closer similarity with H1 rather than H3, whereas the receptor binding properties were more like those of the 1968 H3 virus.
In addition the study found that the avian H2 can bind to both avian and human receptors without mutation. Once in the human body mutations could then improve the ability of the virus to bind to human cells, and as a consequence increase the rate of virus transmission.
“We have established that some avian viruses are more able to gain a foothold in humans because they are already able to bind to human receptors. It is possible that the H1, H2 and H3 strains responsible for the three major pandemics have a propensity for trans-species infection, but it would be useful to extend our investigation to analyse all 16 hemagglutinin subtypes, not least for the purposes of pandemic planning.”Steve Gamblin, Medical Research Council
Structures of receptor complexes formed by hemagglutinins from the Asian Influenza pandemic of 1957, Junfeng Liu, David J. Stevens, Lesley F. Haire, Philip A. Walker, Peter J. Coombs, Rupert J. Russell, Steven J. Gamblin and John J. Skehel, PNAS October 6, 2009 vol. 106 no. 40 17175-17180
DOI: 10.1073/pnas.0906849106
Diamond Light Source is the UK's national synchrotron science facility, located at the Harwell Science and Innovation Campus in Oxfordshire.
Copyright © 2022 Diamond Light Source
Diamond Light Source Ltd
Diamond House
Harwell Science & Innovation Campus
Didcot
Oxfordshire
OX11 0DE
Diamond Light Source® and the Diamond logo are registered trademarks of Diamond Light Source Ltd
Registered in England and Wales at Diamond House, Harwell Science and Innovation Campus, Didcot, Oxfordshire, OX11 0DE, United Kingdom. Company number: 4375679. VAT number: 287 461 957. Economic Operators Registration and Identification (EORI) number: GB287461957003.