Since the Stone Age humans have been adept at selecting materials for specific tasks based on the material’s properties. With the discovery of smelting man learned to manipulate materials to improve performance. Now new materials can be “built” on a nm scale with properties that are required for a host of applications, from solar cells to catalysts in industrial process.
However, producing and characterising these new materials can be a time-consuming process. Dr Jeremy Cockcroft and Dr Jawwad Darr, from University College London, have been using beamline I11 at Diamond to analyse samples made using a novel high throughput hydrothermal synthesis system that can produce new nano materials with slightly varying compositions. Using this new synthesis system, they were able to produce 100 different samples in a single day. It was therefore important that the process of characterising their structures using powder diffraction was equally quick and not a bottleneck, a task for which the robotic sample placement system and fast data collection times on the powder diffraction beamline are ideally suited. A total of 66 specimens were successfully examined in just one day of synchrotron beamtime, providing a wealth of new information regarding their structural properties.
“The new facility at Diamond was used to gain high quality diffraction data on the entire heat treated sample library of nanoceramics. The speed, quantity, and quality of data obtained by this new approach offers an exciting new development which will allow structure-property relationships to be rapidly accessed for nano ceramics and other synthesised materials.”Dr Jeremy Cockcroft, University College London
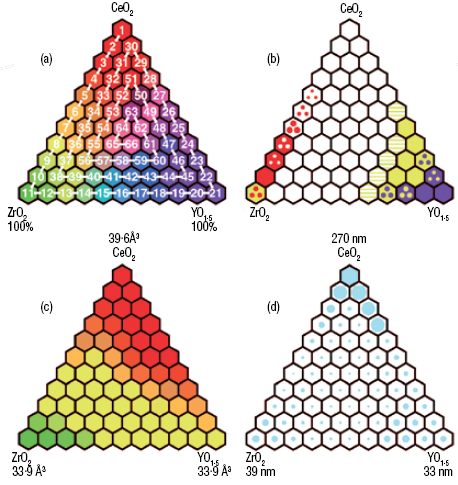 |
| Summary of engineered oxide results with schemes showing: (a) Compositional space for CexZryYzO2-δ (for 0 ≤ x,y,z ≤ 1). Here, each hexagon represents a single sample, and the nominal molar content of Ce, Zr, and Y in each sample is proportional to the amount of red, green, and blue colour, respectively. (b) A simplified phase diagram for CexZryYzO2-δ as deduced from powder diffraction data showing the following phase types: fluorite (grey), monoclinic ZrO2 (yellow), tetragonal ZrO2 (red), bixbyite (blue) and an oxygen-deficient fluorite superstructure (pseudo-Y2O3) with oxygen vacancy ordering (green). Hexagons in a single colour indicate single-phase material, a hexagon with three dots indicates two-phase behavior and those with striped colours indicate a single-phase region. (c) Lattice parameter information obtained from Rietveld analysis converted to mean volume per metal oxide unit showing a smooth pseudo-linear variation with mole fraction. (d) Relative crystallite size (as indicated by the circle diameters) obtained for each of the 66 samples. The largest crystallite size was obtained for pure CeO2 and is shown by a filled hexagon. |
Despite technological advances there are still some areas where the natural world can rival the most advanced engineering materials produced by Man. One such example is the shells of molluscs, which provide protective barriers for the creatures inside. These shells have the strength to withstand the harshness of the environment: crushing actions (from predators, high pressure in the deep ocean and smashing waves) and chemical resistance (saline and fresh water). In addition, the process of production used by these creatures is both robust and environmentally friendly. It is however a process that is still not fully understood.
The method by which these tough nano scale composites are formed has significant implications for the production of tailored bio-materials and other applied fabrics. Understanding the design, synthesis and transformation of these calcium-rich materials is therefore an important component in the development of new bio-medical framework materials (e.g. to encourage bone regeneration) as well as stronger structural composites for other engineering and domestic applications.
The mineral phases in mussel shell are essentially highly stable bi-layers of aragonite on the inside and calcite on the outside shell. Interestingly, the aragonite mineral is not stable in either its manmade or geological form and readily converts to the ubiquitous calcite phase.
Diamond scientists Drs Chiu Tang, Steve Thompson, Julia Parker and Alistair Lennie have studied the transformation from aragonite to calcite in both biogenic mussel shell and man-made samples, which exhibit different structural characteristics to each other during their transformation.
“The ability to manufacture these materials in a form tailored to suit their purpose was first evolved by primitive creatures some 540 million years ago. Our results are adding to our understanding of the structural details in these amazing mineralised assemblies, and may enable us to synthesise new materials with properties that can equal if not better those of the natural world.”Dr Stephen Thompson, Diamond Light Source
Diamond Light Source is the UK's national synchrotron science facility, located at the Harwell Science and Innovation Campus in Oxfordshire.
Copyright © 2022 Diamond Light Source
Diamond Light Source Ltd
Diamond House
Harwell Science & Innovation Campus
Didcot
Oxfordshire
OX11 0DE
Diamond Light Source® and the Diamond logo are registered trademarks of Diamond Light Source Ltd
Registered in England and Wales at Diamond House, Harwell Science and Innovation Campus, Didcot, Oxfordshire, OX11 0DE, United Kingdom. Company number: 4375679. VAT number: 287 461 957. Economic Operators Registration and Identification (EORI) number: GB287461957003.