DNA is constantly under attack. Within the human body there is an army of proteins which can detect when DNA has been damaged and attempt to repair it. One such protein is Helicase XPD.
In humans, mutations in Helicase XPD can lead to a wide range of problems, including developmental defects, hypersensitivity to UV and increased susceptibility to cancer. Scientists from the Centre for Biological Sciences at the University of St Andrews have been using Diamond to determine the structure of this key protein on a molecular scale, to learn about its function and how mutations can lead to such a diverse spectrum of symptoms. This work has been published in the journal Cell.
In Detail
The protein Helicase XPD is a component of the transcription factor IHH (TFIIH). TFIIH plays a dual role, in transcription and repair of DNA. The XPD gene is the target of mutation for patients with xeroderma pigmentosum, trichothio-dystrophy and Cockaynes syndrome, which cause a range of symptoms from brittle hair to increased susceptibility to cancer.
A group of scientists led by Jim Naismith and Malcolm White at the University of St Andrews used Diamond’s I03 beamline to solve the crystal structure of XPD from the crenarchaeon Sulfolobus tokodaii. This provides understanding at the molecular level of the structural basis of helicase activity, and the phenotypes of XPD mutations in humans.
"It is remarkable that this simple structure explains the tragic diseases suffered by those with genetic mutations in the XPD gene. This work was only possible through rapid access to synchrotrons. It was a very difficult piece of work only possible through the team work of all involved."Prof Jim Naismith, University of St Andrews
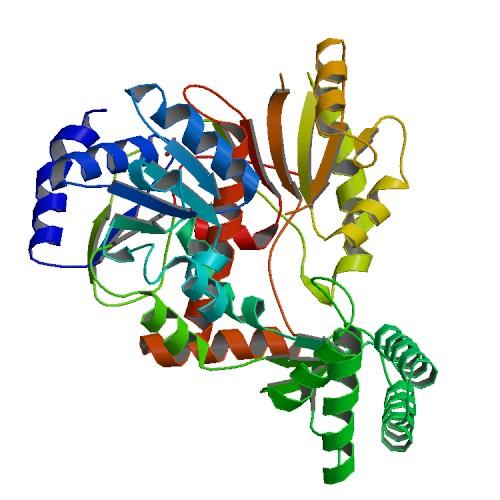
Part of the protein structure of Helicase XPD
Huanting Liu, Jana Rudolf, Kenneth A. Johnson, Stephen A. McMahon, Muse Oke, Lester Carter, Anne-Marie McRobbie, Sara E. Brown, James H. Naismith, and Malcolm F. White, Structure of the DNA Repair Helicase XPD, Cell 133, 801–812, May 30, 2008
DOI 10.1016/j.cell.2008.04.029
Diamond Light Source is the UK's national synchrotron science facility, located at the Harwell Science and Innovation Campus in Oxfordshire.
Copyright © 2022 Diamond Light Source
Diamond Light Source Ltd
Diamond House
Harwell Science & Innovation Campus
Didcot
Oxfordshire
OX11 0DE
Diamond Light Source® and the Diamond logo are registered trademarks of Diamond Light Source Ltd
Registered in England and Wales at Diamond House, Harwell Science and Innovation Campus, Didcot, Oxfordshire, OX11 0DE, United Kingdom. Company number: 4375679. VAT number: 287 461 957. Economic Operators Registration and Identification (EORI) number: GB287461957003.