Bacteria that develop resistance to drugs can cause great problems in the treatment of infections and diseases. Multi-drug resistance bacteria pump the drugs out of their cells through membrane proteins known as transporters. To reveal the structure of these proteins and understand their mechanism it is necessary to isolate the proteins, grow crystals and collect data at powerful X-ray sources. An early success at Diamond Light Source has been achieved with crystals of the multidrug efflux membrane protein AcrB. A native and a substrate bound data sets were collected on beamline I04 to 3.3Å and 3.8Å resolution respectively. The crystals belonged to spacegroup H32 with cell dimensions of (a=b=145.3Å c=519.0Å). The structure was solved using the published AcrB structure1 for molecular replacement. This will provide the groundwork for obtaining the structure of AcrB with other substrates in the translocation channel to shed more information as to the mode of action of this protein.
1 Murakami, S., et al. Nature 419, 587-593, 2002
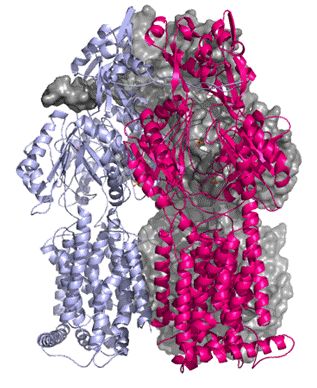 | 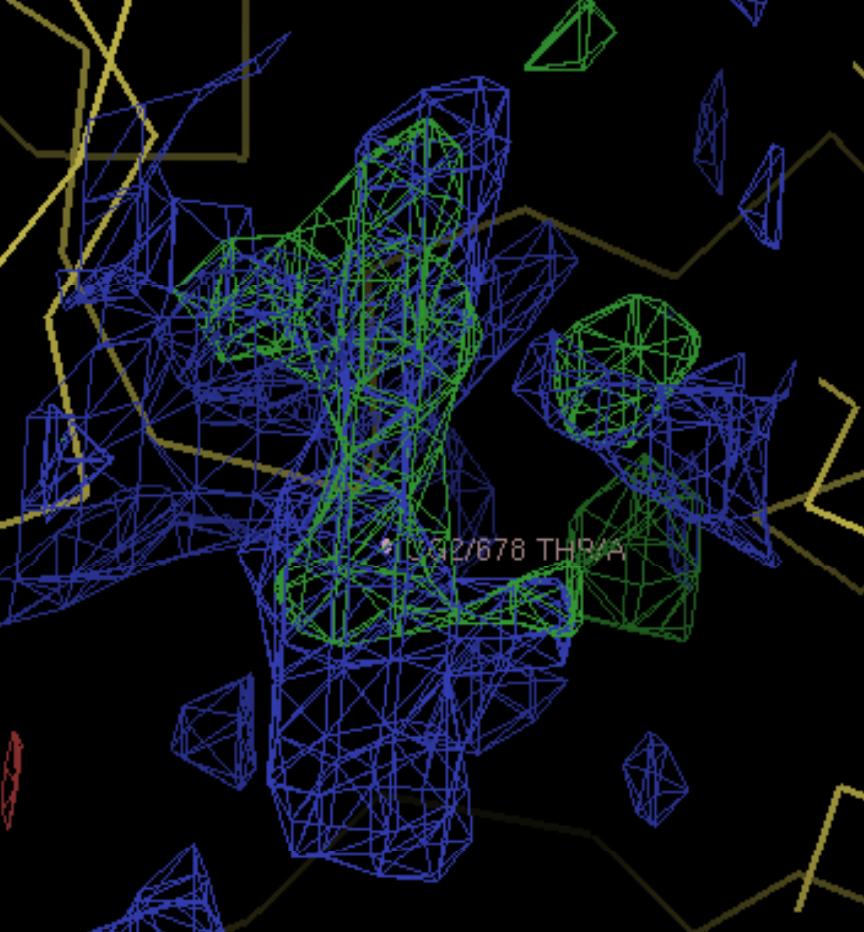 |
| Figure 1. Structure of the trimeric AcrB protein. Ribbon and space-fill representation | Figure 2. 2Fo-Fc (blue) and Fo-Fc (green) density maps after molecular replacement. Density for the substrate is visible |
The structure of the efflux pump AcrB in complex with bile acid, Drew D, Klepsch MM, Newstead S, Flaig R, De Gier JW, Iwata S, Beis K.Molecular Membrane Biology, December 2008; 25(8): 677-682.
PMID: 19023693
Diamond Light Source is the UK's national synchrotron science facility, located at the Harwell Science and Innovation Campus in Oxfordshire.
Copyright © 2022 Diamond Light Source
Diamond Light Source Ltd
Diamond House
Harwell Science & Innovation Campus
Didcot
Oxfordshire
OX11 0DE
Diamond Light Source® and the Diamond logo are registered trademarks of Diamond Light Source Ltd
Registered in England and Wales at Diamond House, Harwell Science and Innovation Campus, Didcot, Oxfordshire, OX11 0DE, United Kingdom. Company number: 4375679. VAT number: 287 461 957. Economic Operators Registration and Identification (EORI) number: GB287461957003.