____________________________________
Industrial Liaison Group:
Tel: +44 (0) 1235 778797
E-mail: [email protected]
Huntington’s disease (HD) is a dominantly-inherited neuropsychiatric disorder which causes severe motor dysfunction, depression and dementia. It has been known since 1993 that the disease is due to mutation of a single gene coding for huntingtin (HTT) that extends the poly-glutamine (poly-Q) repeats in the protein. Inhibiting the enzyme responsible for the cleavage of mutant HTT - thereby reducing the generation of the poly-Q fragments - could be one way of preventing neurodegeneration. Caspase-6 was identified as this processing enzyme, yet, while structures of other caspase enzymes have been studied, caspase-6 has been suggested to be this processing enzyme but, while structures of other caspase enzymes have been solved.
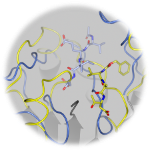
CHDI Foundation, Inc., a biomedical research organization exclusively focused on developing disease-modifying therapies for HD, is working with BioFocus, a drug discovery provider, to solve the structure of the active caspase-6 and in complex with inhibitors, with a view towards developing caspase-6 specific drugs. Using mosquito® Crystal for automated low-volume crystal screening, BioFocus identified crystallization conditions for apo-caspase-6 in the active state (PDB ID 3P45), in complex with the reversible inhibitor Ac-VEID-CHO (PDB ID 3P4U), as well as in complex with the irreversible inhibitor Z-VAD-FMK (PDB ID 3QNW). Optimization of the crystallization conditions of the irreversible inhibitor yielded plate-like crystals typically only 10-20 μm thick, which required synchrotron light to gain sufficient optical resolution.
The team used rapid-access mode to work across several MX beamlines at Diamond, enabling them to solve the structures within a few weeks. They found that surprisingly unlike the reversible inhibitor Ac-VEID-CHO, the irreversible inhibitor Z-VAD-FMK binds to caspase-6 in the inactivated conformational state, and in a binding mode not previously observed for any other caspase-inhibitor complex.
With time scheduled throughout the year for mail-in service data collection, regular customers can have confidence that their data will be collected in a timely fashion. Occasional sample shipments can also be handled with ease, with the team being able to take advantage of any short notice availability of beamtime. With sample arrival to data departure intervals as short as 24 hours, the MX mail-in data collection service can be a valuable addition to any industrial crystallographer’s toolbox.


“The application of Diamond Light Source facilities streamlined our caspase-6 structural biology efforts.”
Dr Alex Kiselyov, Director at CHDI Foundation“The rapid access to the MX stations at the Diamond synchrotron helps us to deliver
final structures to our clients within a few weeks of obtaining the first crystals.”
Dr Ilka Müller, BioFocus

Structure of human caspase-6 in complex with Z-VAD-FMK: New peptide binding mode observed for the non-canonical caspase conformation, I. Müller, M.B. Lamers, A.J. Ritchie, C. Dominguez, I. Munoz-Sanjuan, A. Kiselyov, Bioorg. Med. Chem. Lett., 21, 5244-5247, (2011) (doi:10.1016/j.bmcl.2011.07.041)
Diamond Light Source is the UK's national synchrotron science facility, located at the Harwell Science and Innovation Campus in Oxfordshire.
Copyright © 2022 Diamond Light Source
Diamond Light Source Ltd
Diamond House
Harwell Science & Innovation Campus
Didcot
Oxfordshire
OX11 0DE
Diamond Light Source® and the Diamond logo are registered trademarks of Diamond Light Source Ltd
Registered in England and Wales at Diamond House, Harwell Science and Innovation Campus, Didcot, Oxfordshire, OX11 0DE, United Kingdom. Company number: 4375679. VAT number: 287 461 957. Economic Operators Registration and Identification (EORI) number: GB287461957003.