 | ||
The structures of new ruthenium oxides from aqueous chemistry |
Structures of three new metastable ruthenium oxides have been determined as a result of a collaborative project between three academic institutions and sustainable technology company Johnson Matthey. Ruthenium oxides are used in electro-catalysis for water oxidation and reduction, in heterogeneous catalysis, and are also of interest for their electric and magnetic properties. The majority of transition metal oxides are prepared at high temperatures, resulting in the formation of only the most stable compounds. The potential for many metastable oxides exists, such as compounds with unusual oxidation states that are inaccessible using conventional synthesis conditions. Preparation methods for synthesising such oxides typically require special conditions such as high pressure or strongly oxidising or reducing conditions. By heating the reactants at 200 °C in water in a sealed system, termed hydrothermal synthesis, pressure is generated, resulting in metal oxides that would not be obtainable at high temperatures.
Beamlines I11/B18 Scientific Highlight
Using data collected at the High Resolution Powder Diffraction beamline (I11) and the Core XAFS beamline (B18) at Diamond Light Source, the team characterised the oxides Ca1.5Ru2O7, SrRu2O6 and Ba2Ru3O9(OH). X-ray absorption data collected at B18 allowed determination of the average oxidation states of ruthenium, which is Ru(V) in each case, and magnetisation data were obtained for each material. The materials crystallise directly from solution, suggesting that this route could be an advantageous synthesis method with potential applications in the preparation of other heavy metal oxides, such as those containing Ir(V) or Os(V). This could result in a series of novel materials with interesting magnetic properties without the need for high temperatures.
Ruthenium oxides are currently of widespread interest for their exotic and diverse magnetic and electronic properties (such as high temperature superconductivity in Sr2RuO4 and both metallic conductivity and ferromagnetism in SrRuO3)1,2, and their catalytic activity3. The majority of these oxides contain Ru in the +4 oxidation state, and highly oxidising synthesis conditions are typically required in order to isolate the +5 and +6 oxidation states. The synthesis of ruthenates is currently dominated by the use of high temperature techniques (in excess of 1000 °C), which means that only the most thermodynamically stable of compounds are known. By a simple, low-temperature hydrothermal synthesis route, where inorganic precursors are heated in water under autogeneous pressure, we have succeeded in synthesising three new oxides which contain Ru in the +5 oxidation state. Each of these oxides is found to be metastable and readily decomposes to mixtures of more stable, known Ru(IV) oxides upon heating in air above 300 – 500 °C.
The hydrothermal reaction of potassium perruthenate (KRuO4) and barium peroxide (BaO2) at 200 °C yields Ba2Ru3O9(OH), which adopts a previously unreported crystal structure type. The product was polycrystalline and a single crystal could not be isolated, therefore the structure was solved by direct methods from a combination of powder synchrotron X-ray diffraction (Fig. 1a, collected on I11, and powder neutron diffraction collected at ISIS (Fig. 1b)) . The structure consists of corrugated layers of edge- and corner-sharing RuO6 octahedra interlayed with Ba2+ and H+ ions projecting into the space between the layers (Fig. 1c,d). Bond valence sums and X-ray absorption near edge structure (XANES) spectroscopy, measured on B18, both give an average Ru oxidation state of +5. Magnetometry suggests that the oxide contains Ru(V) ions which ferromagnetically order at 70 K.
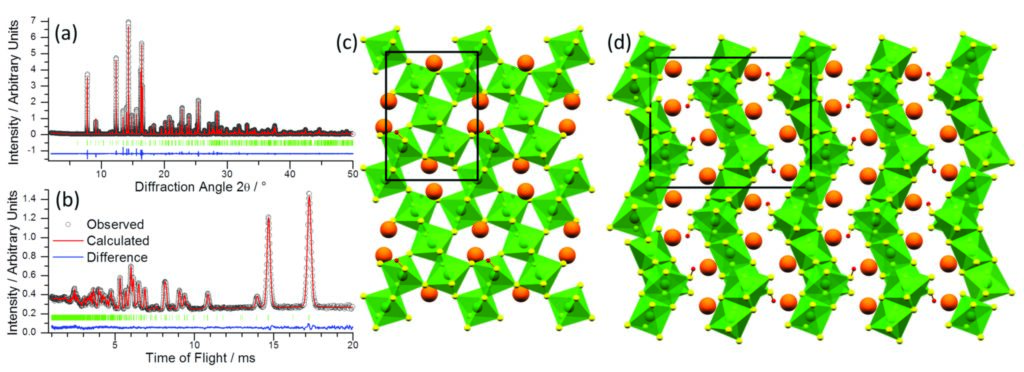
Figure 1: (a) Fitted synchrotron (λ = 0.827154 Å) powder X-ray diffraction pattern and (b) fitted powder neutron diffraction pattern of Ba2Ru3O9(OH). (c) and (d) Structure of Ba2Ru3O9(OH) as viewed in the ab and bc planes, respectively. Ba atoms are shown in orange, H atoms in red, with green RuO6 octahedra.
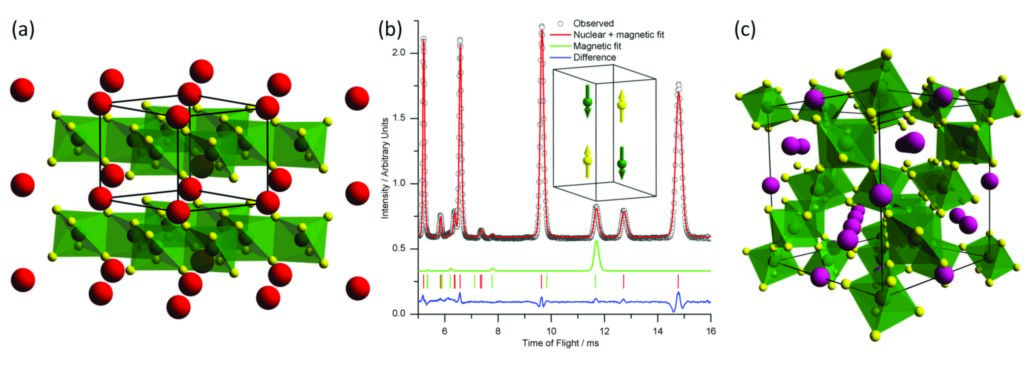
Figure 2: (a) The structure of SrRu2O6. (b) Room temperature powder neutron diffraction pattern of SrRu2O6 with nuclear and magnetic fit. Inset shows the antiferromagnetic ordering of Ru atoms. (c) The structure of Ca1.5Ru2O7. RuO6 octahedra shown in green, Sr atoms in red and Ca atoms in purple.
If strontium peroxide (SrO2) is used instead of BaO2 in a similar reaction, the product is a new oxide SrRu2O6. This adopts a hexagonal structure isomorphous to PbSb2O6 consisting of alternating layers of RuO6 octahedra and SrO6 octahedra (Fig. 2a). Very few examples of magnetic ordering in this structure type exist. Again, bond valence sums and XANES were used to confirm an average Ru oxidation state of +5. Room temperature powder neutron diffraction and magnetometry were used to prove that this material is antiferromagnetically ordered significantly at and above room temperature, a very rare property in a 4d metal oxide. A model for the magnetic structure, fitted to the room temperature neutron diffraction data, consists of antiferromagnetic ordering of the Ru atoms both parallel and perpendicular to the layers (Fig. 2b).

Figure 3: Normalised Ru K-edge XANES spectra of Ca1.5Ru2O7, SrRu2O6 and Ba2Ru3O9(OH) and reference compounds. Inset: a plot of oxidation state as a function of edge position (maximum of 1st derivative).
The analogous reaction using calcium peroxide (CaO2) was found to yield Ca1.5Ru2O7, a highly A-site deficient pyrochlore-type structure, which can be considered a tetrahedral three-dimensional network of corner-sharing RuO6 octahedra with channels of Ca and O running through the network (Fig. 2c). Pyrochlores are of interest as solid electrolytes due to their high anion conductivity and when they contain magnetic ions, they can exhibit magnetic frustration. It was initially unclear as to how the high level of Ca deficiency was tolerated in the structure, since elemental analysis, infrared spectroscopy, thermogravimetric analysis and powder neutron diffraction suggest that there is no accompanying oxide deficiency or hydroxide present. Ru K-edge XANES (Fig. 3), shows that the oxidation state of the Ru is close to +5.5, giving a consistent view of the chemical composition. Measurement of the magnetic susceptibility as a function of temperature found evidence of magnetic frustration in the arrangement of the Ru moments at low temperature.
Source publication:
Hiley, C. I., Lees, M. R., Fisher, J. M., Thompsett, D., Agrestini, S., Smith, R. I. & Walton, R. I. Ruthenium (V) Oxides from Low-Temperature Hydrothermal Synthesis. Angewandte Chemie-International Edition 53, 4423-4427, doi:10.1002/anie.201310110 (2014).
References:
1. Ishida, K. et al. Spin-triplet superconductivity in Sr2RuO4 identified by 17O Knight shift. Nature 396, 658-660, doi:10.1038/25315 (1998).
2. Longo, J. M., Raccah, P. M. & Goodenough, J. B. Magnetic properties of SrRuO3 and CaRuO3. Journal of Applied Physics 39, 1327-1328, doi:10.1063/1.1656282 (1968).
3. Ashcroft, A. T. et al. Selective oxidation of methane to synthesis gas-using transition-metal catalysts. Nature 344, 319-321, doi:10.1038/344319a0 (1990).
Funding acknowledgement:
We thank Johnson Matthey plc for funding via an EPSRC industrial CASE award.
Corresponding author:
Professor Richard Walton, University of Warwick, [email protected]


 A brighter light for science
A brighter light for science
